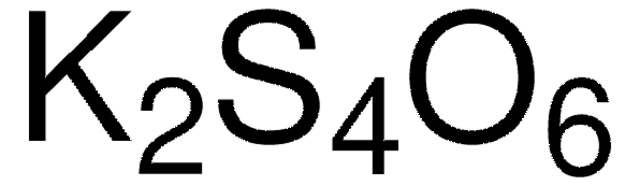S5758
Sodium tetrathionate dihydrate
≥98% (titration)
About This Item
Productos recomendados
Quality Level
assay
≥98% (titration)
solubility
H2O: 200 + 4mL H2O mg, clear to hazy, colorless to faintly yellow
density
2.1 g/mL at 25 °C (lit.)
storage temp.
2-8°C
SMILES string
O.O.[Na+].[Na+].[O-]S(=O)(=O)SSS([O-])(=O)=O
InChI
1S/2Na.H2O6S4.2H2O/c;;1-9(2,3)7-8-10(4,5)6;;/h;;(H,1,2,3)(H,4,5,6);2*1H2/q2*+1;;;/p-2
InChI key
HAEPBEMBOAIUPN-UHFFFAOYSA-L
¿Está buscando productos similares? Visita Guía de comparación de productos
Application
- Gaussia princeps luciferase: a bioluminescent substrate for oxidative protein folding.: Sodium tetrathionate dihydrate is used to study its impact on the oxidative folding of proteins, utilizing Gaussia princeps luciferase as a bioluminescent substrate. This study advances understanding of protein chemistry and bioluminescence applications in biochemical assays (Yu T et al., 2018).
- A spectroscopic investigation into the reaction of sodium tetrathionate with cysteine.: The research utilizes spectroscopic methods to study the reaction between sodium tetrathionate dihydrate and cysteine, providing insights into the chemical interactions and potential applications in biochemistry and molecular biology (Church JS et al., 2008).
https://doi.org/10.2174/092986612800190973
https://doi.org/10.1080/10826068.2011.544230
https://doi.org/10.11150/kansenshogakuzasshi.83.380
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico