T71803
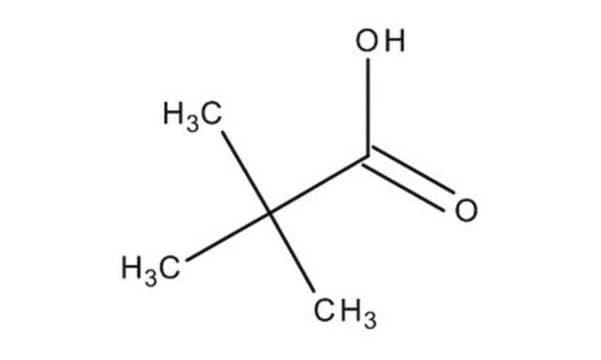
Pivalic acid
99%
Sinónimos:
2,2-Dimethylpropionic acid, Trimethylacetic acid
About This Item
Productos recomendados
vapor density
3.6 (vs air)
Quality Level
vapor pressure
9.75 mmHg ( 60 °C)
assay
99%
reaction suitability
reaction type: C-H Activation
bp
163-164 °C (lit.)
mp
32-35 °C (lit.)
density
0.889 g/mL at 25 °C (lit.)
functional group
carboxylic acid
SMILES string
OC(C(C)(C)C)=O
InChI
1S/C5H10O2/c1-5(2,3)4(6)7/h1-3H3,(H,6,7)
InChI key
IUGYQRQAERSCNH-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
General description
Application
- As a co-catalyst with palladium for the arylation of unactivated arenes and N-heterocycles.
- As an additive to facilitate the carbonylative suzuki reactions to synthesize biaryl ketones from aryl iodides and arylboronic acids by using palladium nanoparticles as catalyst.
- In the cyclization reaction of benzamides with alkynes to synthesize isoquinolones in the presence of 8-aminoquinoline ligand and cobalt catalyst.
Caution
related product
signalword
Warning
hcodes
Hazard Classifications
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2
Storage Class
11 - Combustible Solids
wgk_germany
WGK 1
flash_point_f
147.2 °F - closed cup
flash_point_c
64 °C - closed cup
ppe
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico