357774
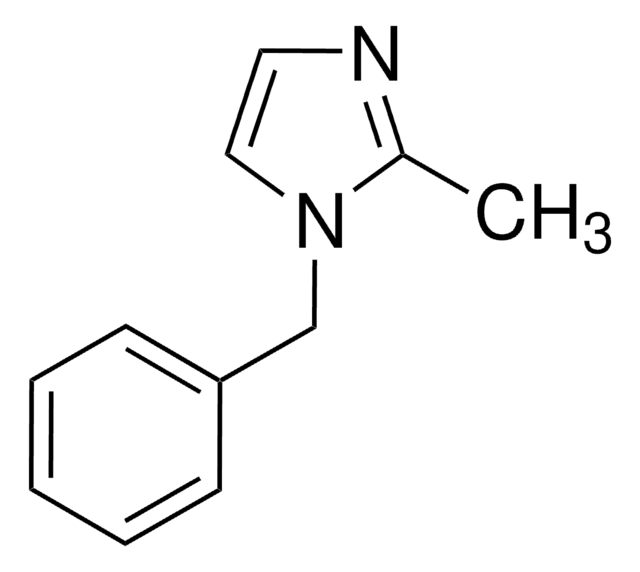
1-Phenylimidazole
97%
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula empírica (notación de Hill):
C9H8N2
Número de CAS:
Peso molecular:
144.17
Número MDL:
Código UNSPSC:
12352005
ID de la sustancia en PubChem:
NACRES:
NA.22
Productos recomendados
Nivel de calidad
Ensayo
97%
Formulario
liquid
bp
142 °C/15 mmHg (lit.)
mp
13 °C (lit.)
densidad
1.14 g/mL at 25 °C (lit.)
cadena SMILES
c1ccc(cc1)-n2ccnc2
InChI
1S/C9H8N2/c1-2-4-9(5-3-1)11-7-6-10-8-11/h1-8H
Clave InChI
SEULWJSKCVACTH-UHFFFAOYSA-N
Descripción general
1-Phenylimidazole is an imidazole derivative. It induces 7-ethoxyresorufin-O-deethylase (EROD) activity in rainbow trout (Oncorhynchus mykiss) hepatocytes. The S(1)→S(0) transition of 1-phenylimidazole has been investigated in a supersonic jet expansion by resonant two-photon ionization. 1-Phenylimidazole is reported to be inhibitor of calmodulin-dependent nitric-oxide synthase from bovine brain and GHs pituitary cells.
Aplicación
1-Phenylimidazole is a suitable reagent used to investigate its effect on the citrulline formation by bovine brain nitric-oxide synthase.
Palabra de señalización
Warning
Frases de peligro
Clasificaciones de peligro
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Órganos de actuación
Respiratory system
Código de clase de almacenamiento
10 - Combustible liquids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
235.4 °F - closed cup
Punto de inflamabilidad (°C)
113 °C - closed cup
Equipo de protección personal
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Priyadarshini Balaraman et al.
Biochimica et biophysica acta. General subjects, 1863(2), 304-312 (2018-11-06)
The camphor-degrading microorganism, Pseudomonas putida strain ATCC 17453, is an aerobic, gram-negative soil bacterium that uses camphor as its sole carbon and energy source. The genes responsible for the catabolic degradation of camphor are encoded on the extra-chromosomal CAM plasmid.
A J Green et al.
Journal of biological inorganic chemistry : JBIC : a publication of the Society of Biological Inorganic Chemistry, 6(5-6), 523-533 (2001-07-27)
The bioI gene has been sub-cloned and over-expressed in Escherichia coli, and the protein purified to homogeneity. The protein is a cytochrome P450, as indicated by its visible spectrum (low-spin haem iron Soret band at 419 nm) and by the
Filippo Monti et al.
Inorganic chemistry, 54(6), 3031-3042 (2015-03-06)
A series of cationic iridium(III) complexes with two carbene-based cyclometalating ligands and five different N^N bipyridine and 1,10-phenanthroline ancillary ligands is presented. For the first time--in the frame of a rarely studied class of bis(heteroleptic) iridium complexes with two carbene-based
A Jos et al.
Toxicology in vitro : an international journal published in association with BIBRA, 21(7), 1307-1310 (2007-05-25)
The classical pathway for induction of cytochrome P4501A (CYP1A) by xenobiotics is ligand binding to the aryl hydrocarbon receptor (AhR). However, several studies with mammalian cell systems point out a range of xenobiotics including imidazole derivatives, which are able to
M R Anari et al.
Chemical research in toxicology, 9(6), 924-931 (1996-09-01)
Organic hydroperoxides are believed to be primarily detoxified in cells by the GSH peroxidase/GSSG reductase system and activated to cytotoxic radical species by non-heme iron. However, organic hydroperoxides seem to be bioactivated by cytochrome P450 (P450) in isolated hepatocytes as
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico











![1-[2-(Trifluoromethyl)phenyl]imidazole](/deepweb/assets/sigmaaldrich/product/structures/150/780/ea7e6b25-7659-422e-868c-8df7fd70d66e/640/ea7e6b25-7659-422e-868c-8df7fd70d66e.png)

