103012
3-Hydroxy-4-methoxycinnamic acid, predominantly trans
97%
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula lineal:
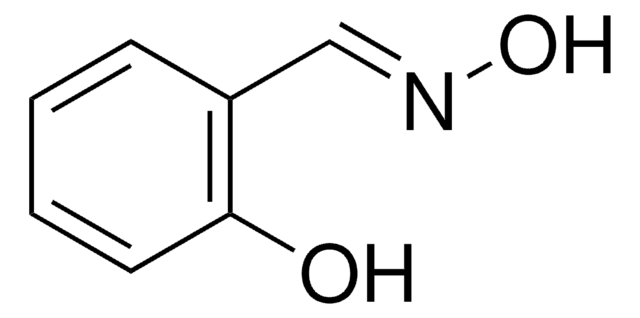
HOC6H3(OCH3)CH=CHCO2H
Número de CAS:
Peso molecular:
194.18
Número CE:
Número MDL:
Código UNSPSC:
12352100
ID de la sustancia en PubChem:
NACRES:
NA.22
Productos recomendados
Ensayo
97%
Formulario
solid
mp
230 °C (dec.) (lit.)
grupo funcional
carboxylic acid
cadena SMILES
COc1ccc(\C=C\C(O)=O)cc1O
InChI
1S/C10H10O4/c1-14-9-4-2-7(6-8(9)11)3-5-10(12)13/h2-6,11H,1H3,(H,12,13)/b5-3+
Clave InChI
QURCVMIEKCOAJU-HWKANZROSA-N
Categorías relacionadas
Descripción general
3-Hydroxy-4-methoxycinnamic acid, predominantly trans is available as white crystals. 3-Hydroxy-4-methoxycinnamic acid is isolated from the aerial part of Artemisia capillaris, Chinese medicinal plant. It is a component of chinese herbal medicine used for a pain killer and stomachic. It is an efficient acetylcholine inhibitor. 3-Hydroxy-4-methoxycinnamic acid is bioactive metabolite of Spilanthes acmella Murr. It increases the resistance of low-density lipoprotein (LDL) to oxidation.
Aplicación
3-Hydroxy-4-methoxycinnamic acid was used in the synthesis of tranilast and various tranilast analogs (cinnamoyl anthranilates) by genetically engineered Saccharomyces cerevisiae yeast strain. It was also used in the synthesis of glycoside compounds by undergoing glycosidation.
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 2
Equipo de protección personal
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
Certificados de análisis (COA)
Lot/Batch Number
¿No ve la versión correcta?
Si necesita una versión concreta, puede buscar un certificado específico por el número de lote.
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Supaluk Prachayasittikul et al.
Molecules (Basel, Switzerland), 14(2), 850-867 (2009-03-04)
Spilanthes acmella Murr. (Compositae) has been used as a traditional medicine for toothache, rheumatism and fever. Its extracts had been shown to exhibit vasorelaxant and antioxidant activities. Herein, its antimicrobial, antioxidant and cytotoxic activities were evaluated. Agar dilution method assays
I-Min Liu et al.
The Journal of pharmacology and experimental therapeutics, 307(3), 1196-1204 (2003-09-17)
We investigated the mechanism(s) by which isoferulic acid lowers plasma glucose levels in streptozotocin-induced diabetic rats (STZ-diabetic rats). In STZ-diabetic rats, isoferulic acid dose dependently lowered plasma glucose concentrations and increased plasma beta-endorphin-like immunoreactivity (BER). Both of these effects of
Mariana Monteiro et al.
The Journal of nutrition, 137(10), 2196-2201 (2007-09-22)
Chlorogenic acids (CGA) are abundant phenolic compounds in coffee, with caffeoylquinic (CQA), feruloylquinic (FQA), and dicaffeoylquinic (diCQA) acids being the major subclasses. Despite the potential biopharmacological properties attributed to these compounds, little is known about their bioavailability in humans. In
I M Liu et al.
British journal of pharmacology, 129(4), 631-636 (2000-02-22)
Wistar rats with streptozotocin-induced diabetes (STZ-diabetic rats), which is similar to human insulin-dependent diabetic mellitus (IDDM), were employed to investigate the antihyperglycemic action of isoferulic acid. A single intravenous injection of isoferulic acid decreased the plasma glucose in a dose-dependent
Adriana Farah et al.
The Journal of nutrition, 138(12), 2309-2315 (2008-11-22)
Chlorogenic acids (CGA) are cinnamic acid derivatives with biological effects mostly related to their antioxidant and antiinflammatory activities. Caffeoylquinic acids (CQA) and dicaffeoylquinic acids (diCQA) are the main CGA found in nature. Because green coffee is a major source of
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico