All Photos(1)
About This Item
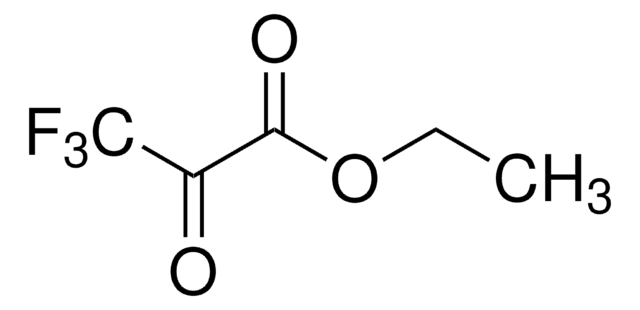
Linear Formula:
CF3COCO2CH3
CAS Number:
Molecular Weight:
156.06
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
97%
refractive index
n20/D 1.332 (lit.)
bp
86 °C (lit.)
density
1.529 g/mL at 25 °C (lit.)
functional group
ester
fluoro
ketone
SMILES string
COC(=O)C(=O)C(F)(F)F
InChI
1S/C4H3F3O3/c1-10-3(9)2(8)4(5,6)7/h1H3
InChI key
XGLLQDIWQRQROJ-UHFFFAOYSA-N
Related Categories
General description
Methyl 3,3,3-trifluoropyruvate is an alkyl 3,3,3-trifluoropyruvate that can be synthesized using hexafluoropropene-1,2-oxide (HFPO) as a starting material. It reacts with aromatic amines, benzylic manoamines and diamines to form the corresponding hemiaminals.
Application
Methyl 3,3,3-trifluoropyruvate may be used in the synthesis of:
- 2-hydroxy-2-trifluoromethylbutan-4-olides
- 2-(trifluoromethyl)butan-4-olides
- 4-trifluoromethyl-(2H)-pyridazin-3-ones
- methyl 3-methoxy-2-trifluoromethylacrylate
Signal Word
Warning
Hazard Statements
Hazard Classifications
Flam. Liq. 3
Storage Class Code
3 - Flammable liquids
WGK
WGK 3
Flash Point(F)
78.8 °F - closed cup
Flash Point(C)
26 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Fluorinated butanolides and butenolides: Part 8. 2-(Trifluoromethyl) butan-4-olides by synthesis from methyl 3, 3, 3-trifluoropyruvate as building block.
Paleta O, et al.
Journal of Fluorine Chemistry, 111(2), 175-184 (2001)
A convenient synthesis of 4-trifluoromethyl-(2H)-pyridazin-3-ones from methyl 3, 3, 3-trifluoropyruvate.
Sibgatulin DA, et al.
Synlett, 12, 1907-1911 (2005)
Methyl 3,3,3-trifluoropyruvate: an improved procedure starting from hexafluoropropene-1, 2-oxide; identification of byproducts.
Dolensky B, et al.
Journal of Fluorine Chemistry, 115(1), 67- 74 (2002)
Fluorinated butanolides and butenolides: Part 9. Synthesis of 2-(trifluoromethyl) butan-4-olides by Wittig reaction using methyl 3, 3, 3-trifluoropyruvate.
Palecek J, et al.
Journal of Fluorine Chemistry, 113(2), 177-183 (2002)
Methyl 3, 3, 3-trifluoropyruvate hemiaminals: Stability and transaminations.
Dolensky B, et al.
Journal of Fluorine Chemistry, 126(5), 745-751 (2005)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service