Select a Size
| Size/SKU | Availability | Price |
|---|---|---|
100 mg | Please contact Customer Service for Availability | €118.00 |
250 mg | Please contact Customer Service for Availability | €187.00 |
1 g | Please contact Customer Service for Availability | €590.00 |
About This Item
Quality Segment
form
powder or crystals
antibiotic activity spectrum
Gram-negative bacteria, Gram-positive bacteria
mode of action
cell wall synthesis | interferes
storage temp.
2-8°C
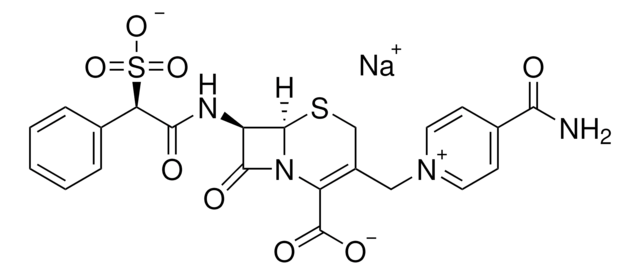
SMILES string
[Na+].NC(=O)c1cc[n+](CC2=C(N3[C@H](SC2)[C@H](NC(=O)[C@@H](c4ccccc4)S([O-])(=O)=O)C3=O)C([O-])=O)cc1
InChI
1S/C22H20N4O8S2.Na/c23-18(27)13-6-8-25(9-7-13)10-14-11-35-21-15(20(29)26(21)16(14)22(30)31)24-19(28)17(36(32,33)34)12-4-2-1-3-5-12;/h1-9,15,17,21H,10-11H2,(H4-,23,24,27,28,30,31,32,33,34);/q;+1/p-1/t15-,17-,21-;/m1./s1
InChI key
REACMANCWHKJSM-DWBVFMGKSA-M
General description
Application
Biochem/physiol Actions
Other Notes
1 of 1
This Item | |||
|---|---|---|---|
| mode of action cell wall synthesis | interferes | mode of action cell wall synthesis | interferes | mode of action cell wall synthesis | interferes | mode of action cell wall synthesis | interferes |
| antibiotic activity spectrum Gram-negative bacteria, Gram-positive bacteria | antibiotic activity spectrum Gram-negative bacteria, Gram-positive bacteria | antibiotic activity spectrum Gram-negative bacteria, Gram-positive bacteria | antibiotic activity spectrum Gram-negative bacteria, Gram-positive bacteria |
| Quality Level 200 | Quality Level 200 | Quality Level 200 | Quality Level 200 |
| form powder or crystals | form powder | form powder or crystals | form powder |
| storage temp. 2-8°C | storage temp. 2-8°C | storage temp. 2-8°C | storage temp. 2-8°C |
Storage Class
11 - Combustible Solids
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Faceshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.