Sign In to View Organizational & Contract Pricing.
Select a Size
Change View
| Pack Size | SKU | Availability | Price |
|---|---|---|---|
| 100 mg | Available to ship TODAYfromMILWAUKEE | $755.00 |
About This Item
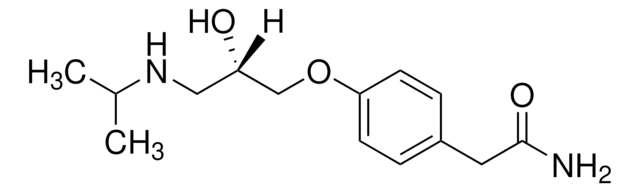
Linear Formula:
(CH3)2CHNHCH2CH(OH)CH2OC6H4CH2CONH2
CAS Number:
Molecular Weight:
266.34
NACRES:
NA.77
PubChem Substance ID:
UNSPSC Code:
12352200
MDL number:
Form:
powder
form
powder
Quality Segment
optical activity
[α]23/D −10.5°, c = 0.5 in H2O(lit.)
color
white to beige
mp
148-152 °C (lit.)
solubility
0.1 M HCl: soluble
SMILES string
CC(C)NC[C@H](O)COc1ccc(CC(N)=O)cc1
InChI
1S/C14H22N2O3/c1-10(2)16-8-12(17)9-19-13-5-3-11(4-6-13)7-14(15)18/h3-6,10,12,16-17H,7-9H2,1-2H3,(H2,15,18)/t12-/m0/s1
InChI key
METKIMKYRPQLGS-LBPRGKRZSA-N
Gene Information
human ... ADRB1(153)
Application
(S)-(-)-Atenolol may be used as a standard in the preparation of enantiomer for high-performance liquid chromatography-tandem mass spectrometry studies (HPLC-MS/MS).[1] It has also been used in the β blocking molecule for Raman spectroscopy studies.
Biochem/physiol Actions
(S)-(-)-Atenolol is a β1-adrenergic receptor antagonist and an active enantiomer of atenolol.[1]
1 of 1
This Item | |||
|---|---|---|---|
| form powder | form - | form - | form powder |
| Quality Level 100 | Quality Level - | Quality Level - | Quality Level 300 |
| solubility 0.1 M HCl: soluble | solubility - | solubility - | solubility - |
| color white to beige | color - | color - | color - |
| mp 148-152 °C (lit.) | mp - | mp - | mp - |
| optical activity [α]23/D −10.5°, c = 0.5 in H2O(lit.) | optical activity - | optical activity - | optical activity - |
Still not finding the right product?
Explore all of our products under (S)-(−)-Atenolol
Storage Class
11 - Combustible Solids
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Related Content
Global Trade Item Number
| SKU | GTIN |
|---|---|
| A143-25MG | 04061832268934 |
| A143-100MG | 04061833342336 |
| A143-10MG | 04061832088570 |