All Photos(1)
About This Item
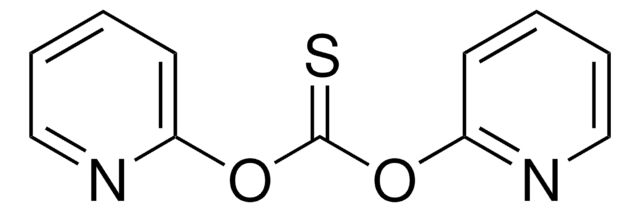
Empirical Formula (Hill Notation):
C11H8N2O2S
CAS Number:
Molecular Weight:
232.26
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
assay
97%
mp
163-166 °C (lit.)
SMILES string
O=C1C=CC=CN1C(=S)N2C=CC=CC2=O
InChI
1S/C11H8N2O2S/c14-9-5-1-3-7-12(9)11(16)13-8-4-2-6-10(13)15/h1-8H
InChI key
KXMMNJQMGILZDB-UHFFFAOYSA-N
Related Categories
Application
1,1′-Thiocarbonyldi-2(1H)-pyridone was used in the preparation of:
- thio-analogs of thioureas
- sulforaphane
- 2-furan-2-yl-3-hydroxy-6-isothiocyanato-chromen-4-one
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
C Clifford Conaway et al.
Cancer research, 65(18), 8548-8557 (2005-09-17)
We have shown previously that naturally occurring isothiocyanates derived from cruciferous vegetables and their N-acetylcysteine conjugates inhibit lung adenoma formation induced by tobacco carcinogens in A/J mice at the post-initiation stage. The tumor-inhibitory activity by these compounds is linked with
Andrey S Klymchenko et al.
The journal of physical chemistry. B, 112(38), 12050-12055 (2008-09-05)
Herein, the efficient interaction of an environment-sensitive fluorophore that undergoes excited-state intramolecular proton transfer (ESIPT) with DNA has been realized by conjugation of a 3-hydroxychromone (3HC) with polycationic spermine. On binding to a double-stranded DNA (dsDNA), the ratio of the
Sun-Young Jang et al.
Bioorganic & medicinal chemistry letters, 14(15), 3881-3883 (2004-07-01)
A series of 4-arylpiperazin-1-yl-3-phenyloxazolidin-2-one derivatives with diversification of the N-substituents such as methylene O-linked heterocycles, thioamide, dithiocarbamate, thiourea, and thiocarbamate were synthesized and evaluated as antibacterial agents. Their in vitro activities (MIC) were evaluated against MRSA and VRE resistant Gram-positive
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service