126233
4,6-Dihydroxy-5-nitropyrimidine
95%
Synonym(s):
5-Nitro-4,6-pyrimidinediol
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C4H3N3O4
CAS Number:
Molecular Weight:
157.08
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
Recommended Products
assay
95%
form
solid
mp
>300 °C (lit.)
functional group
nitro
SMILES string
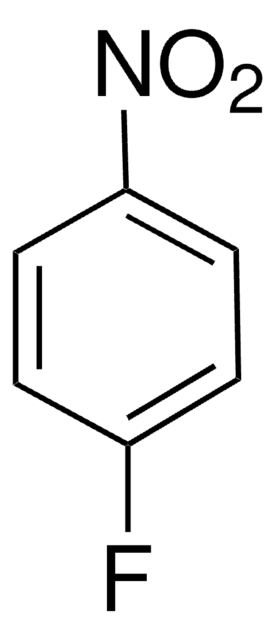
Oc1ncnc(O)c1[N+]([O-])=O
InChI
1S/C4H3N3O4/c8-3-2(7(10)11)4(9)6-1-5-3/h1H,(H2,5,6,8,9)
InChI key
ABTLZAVJDRUDNG-UHFFFAOYSA-N
Biochem/physiol Actions
4, 6-dihydroxy-5-nitropyrimidine is an inhibitor of thymidine phosphorylase activity. It is also a potent and selective inhibitor of 4-nitrophenol glucuronidation.
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
E Miszczak-Zaborska et al.
Zeitschrift fur Naturforschung. C, Journal of biosciences, 52(9-10), 670-675 (1997-11-28)
Partially purified samples of thymidine phosphorylase were obtained from four preparations of human uterine leiomyomas and uteri using the method of Yoshimura et al. (1990), Biochim. Biophys. Acta 1034, 107-113. Among the studied twelve pyrimidine derivatives, 5-bromouracil, 5-nitrouracil, 5-fluorouracil, 6-aminouracil
Z Naydenova et al.
Comparative biochemistry and physiology. Part C, Pharmacology, toxicology & endocrinology, 112(3), 321-325 (1995-11-01)
Thirty-one differently substituted pyrimidine bases were tested for their inhibitory effect on the glucuronidation of 4-nitrophenol and phenolphthalein by rat liver microsomes. 5-Nitrouracil (compound 1) and its isomer 4,6-dihydroxy-5-nitropyrimidine (compound 2) were the most potent and selective inhibitors of 4-nitrophenol
Andrew C Kotze et al.
Antimicrobial agents and chemotherapy, 58(12), 7475-7483 (2014-10-08)
We used an enzyme induction approach to study the role of detoxification enzymes in the interaction of the anthelmintic compound naphthalophos with Haemonchus contortus larvae. Larvae were treated with the barbiturate phenobarbital, which is known to induce the activity of
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service