A13204
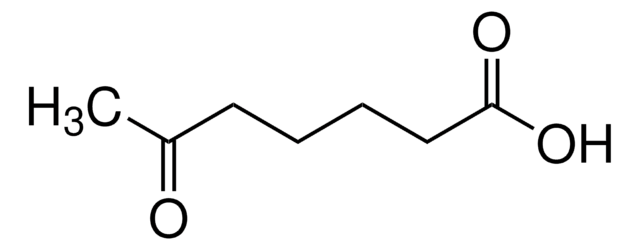
4-Acetylbutyric acid
97%
Synonym(s):
5-Ketohexanoic acid
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
CH3CO(CH2)3CO2H
CAS Number:
Molecular Weight:
130.14
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
97%
form
liquid
refractive index
n20/D 1.4451 (lit.)
bp
274-275 °C (lit.)
mp
13-14 °C (lit.)
density
1.09 g/mL at 25 °C (lit.)
SMILES string
CC(=O)CCCC(O)=O
InChI
1S/C6H10O3/c1-5(7)3-2-4-6(8)9/h2-4H2,1H3,(H,8,9)
InChI key
MGTZCLMLSSAXLD-UHFFFAOYSA-N
Application
4-Acetylbutyric acid may be used in the preparation of the following compounds:
- 5-hydroxyhexanoic acid
- 6-methyl1-3,4-dihydro-pyran-2-one, precursor for 5-acetyl-tetrahydro-2-(3H)-furanone
- substituted N-aminolactams
- (±)-5-methyl-δ-valerolactone
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
A versatile and concise route to functionally substituted ?-butyrolactones and spiro-XXX-butyrolactones (lactone annelation)
Mandal AK and Jawalkar DG
Tetrahedron Letters, 27.1, 99-100 (1986)
Identification of 5-hydroxyhexanoic acid, 4-hydroxyheptanoic acid and 4-hydroxyoctanoic acid as new constituents of bacterial polyhydroxyalkanoic acids.
Valentin HE, et al.
Applied Microbiology and Biotechnology, 46(3), 261-267 (1996)
Jie Yao et al.
Water science and technology : a journal of the International Association on Water Pollution Research, 79(11), 2195-2202 (2019-07-19)
The monoterpene alcohol α-terpineol is extensively used as the foaming agent in mineral processing and can be released to environment along with the wastewater. This study evaluated the feasibility of eliminating α-terpineol in water by ultraviolet irradiation (UV) in combination
Jaroslav Kríz et al.
The journal of physical chemistry. B, 111(22), 6118-6126 (2007-05-15)
Experimental and theoretical evidence for the proximity effect as a basic mechanism of the hydrogen bond cooperativity was obtained in a model system. Hydrogen bond (HB) interaction between poly(4-vinylpyridine) (PVP) and selected acids as HB donors was studied using PFG
(2S, 6S, 8S)-2, 8-Dimethyl-1, 7-dioxaspiro [5.5] undecane: A natural spiroacetal lacking anomeric stabilisation
Chen J, et al.
Tetrahedron Asymmetry, 6.4, 967- 972 (1995)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service