Przejdź do
Wybierz wielkość
| Do Państwa/SKU | Dostępność | Cena netto |
|---|---|---|
10 mg | Skontaktuj się z Obsługą Klienta, aby uzyskać informacje na temat dostępności | 568,00 zł |
50 mg | Skontaktuj się z Obsługą Klienta, aby uzyskać informacje na temat dostępności | 2280,00 zł 1938,00 zł |
Informacje o tej pozycji
568,00 zł
Nazwa produktu
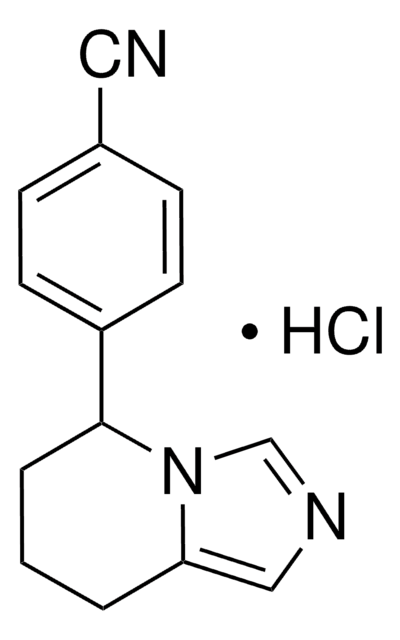
Fadrozole hydrochloride, ≥98% (HPLC)
Quality Segment
assay
≥98% (HPLC)
form
powder
solubility
DMSO: >20 mg/mL
originator
Novartis
storage temp.
room temp
SMILES string
Cl.N#Cc1ccc(cc1)C2CCCc3cncn23
InChI
1S/C14H13N3.ClH/c15-8-11-4-6-12(7-5-11)14-3-1-2-13-9-16-10-17(13)14;/h4-7,9-10,14H,1-3H2;1H
InChI key
UKCVAQGKEOJTSR-UHFFFAOYSA-N
Biochem/physiol Actions
Features and Benefits
1 of 1
Ta pozycja | |||
|---|---|---|---|
| assay ≥98% (HPLC) | assay ≥98% (HPLC) | assay ≥98% (HPLC) | assay ≥98% (HPLC) |
| form powder | form powder | form powder | form powder |
| Quality Level 100 | Quality Level 100 | Quality Level 100 | Quality Level 100 |
| storage temp. room temp | storage temp. room temp | storage temp. room temp | storage temp. room temp |
| solubility DMSO: >20 mg/mL | solubility DMSO: 20 mg/mL, clear | solubility DMSO: 10 mg/mL, clear | solubility DMSO: 2 mg/mL, clear (warmed) |
| originator Novartis | originator - | originator - | originator Johnson & Johnson |
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 3 Oral - Repr. 2
Klasa składowania
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
wgk
WGK 2
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Faceshields, Gloves
Wybierz jedną z najnowszych wersji:
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.