Select a Size
| Size/SKU | Availability | Price |
|---|---|---|
1 mg | Please contact Customer Service for Availability | PLN 876.00 |
5 mg | Please contact Customer Service for Availability | PLN 3,260.00 PLN 2,771.00 |
10 mg | Please contact Customer Service for Availability | PLN 5,760.00 |
About This Item
Product Name
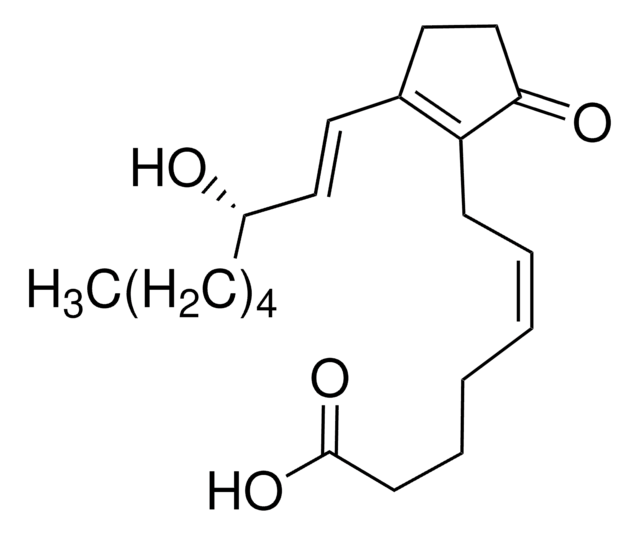
Prostaglandin D2, ≥95%, synthetic
biological source
synthetic
Quality Segment
assay
≥95%
form
powder
storage temp.
−20°C
SMILES string
[H][C@]1(C\C=C/CCCC(O)=O)[C@@H](O)CC(=O)[C@]1([H])\C=C\[C@@H](O)CCCCC
InChI
1S/C20H32O5/c1-2-3-6-9-15(21)12-13-17-16(18(22)14-19(17)23)10-7-4-5-8-11-20(24)25/h4,7,12-13,15-18,21-22H,2-3,5-6,8-11,14H2,1H3,(H,24,25)/b7-4-,13-12+/t15-,16+,17+,18-/m0/s1
InChI key
BHMBVRSPMRCCGG-OUTUXVNYSA-N
Biochem/physiol Actions
Features and Benefits
1 of 1
This Item | |||
|---|---|---|---|
| assay ≥95% | assay ≥98% | assay ≥98% | assay ≥98% (HPLC) |
| form powder | form powder | form solution | form (powder to crystals) |
| Quality Level 100 | Quality Level 200 | Quality Level 100 | Quality Level 200 |
| storage temp. −20°C | storage temp. −20°C | storage temp. −20°C | storage temp. −20°C |
| biological source synthetic | biological source synthetic | biological source synthetic | biological source synthetic |
Still not finding the right product?
Explore all of our products under Prostaglandin D2
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 4 Oral - Repr. 1B
Storage Class
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type P3 (EN 143) respirator cartridges
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.