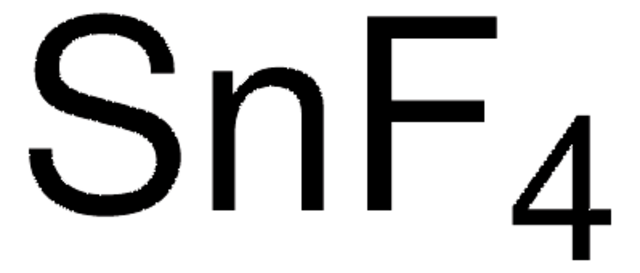14520
Tin(IV) chloride
fuming, ≥99%
Synonym(s):
Stannic chloride
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
SnCl4
CAS Number:
Molecular Weight:
260.52
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
Recommended Products
Quality Level
Assay
≥99%
reaction suitability
reagent type: catalyst
core: tin
density
2.217 g/mL at 20 °C (lit.)
anion traces
sulfate (SO42-): ≤100 mg/kg
cation traces
As: ≤50 mg/kg
Fe: ≤10 mg/kg
SMILES string
Cl[Sn](Cl)(Cl)Cl
InChI
1S/4ClH.Sn/h4*1H;/q;;;;+4/p-4
InChI key
HPGGPRDJHPYFRM-UHFFFAOYSA-J
Looking for similar products? Visit Product Comparison Guide
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Aquatic Chronic 3 - Eye Dam. 1 - Skin Corr. 1B - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 1
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Gerry A Griffith et al.
Journal of the American Chemical Society, 128(40), 13130-13141 (2006-10-05)
Difluorinated alkenoate ethyl 3,3-difluoro-2-(N,N-diethylcarbamoyloxy)-2-propenoate reacts rapidly and in high yield with furan and a range of substituted furans in the presence of a tin(IV) catalyst. Non-fluorinated congener 2-(N,N-diethylcarbamoyloxy)-2-propenoate fails to react at all under the same conditions. These reactions have
Yasuyuki Kita et al.
The Journal of organic chemistry, 71(14), 5191-5197 (2006-07-01)
The Lewis acid-promoted rearrangement of 2,2,3,3-tetrasubstituted 2,3-epoxy alcohols with several kinds of protecting groups was investigated. When SnCl4 is used as a Lewis acid, the reaction proceeds in a regio- and stereo-controlled manner to afford two types of carbonyl compounds
Raquel Coronel et al.
Molecular neurobiology, 56(2), 1248-1261 (2018-06-09)
Amyloid precursor protein (APP) is implicated in neural development as well as in the pathology of Alzheimer's disease (AD); however, its biological function still remains unclear. It has been reported that APP stimulates the proliferation and neuronal differentiation of neural
Colin O'Brien et al.
Chemistry (Weinheim an der Bergstrasse, Germany), 13(3), 902-909 (2006-11-07)
The reaction of silylated nucleophiles with 6,1-anhydroglucopyranuronic acid (glucuronic acid 6,1-lactones) catalysed by tin(IV) chloride provides 1,2-trans or 1,2-cis (deoxy)glycosides in a manner dependent on the donor structure. The alpha-glycoside was obtained for reactions of the donor with the 2-acyl
Gary B Evans et al.
The Journal of organic chemistry, 69(6), 2217-2220 (2004-04-03)
Promotion by Lewis acid of the addition of some aryllithiums to a carbohydrate-based imine, which has allowed a more facile synthesis of some imino-C-nucleoside analogues, is described. Use of the corresponding nitrone does not assist in some cases, but lithiated
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service