MAB351R
Anti-Glutamate Decarboxylase Antibody, 65 kDa isoform, clone GAD-6
clone GAD-6, Chemicon®, from mouse
Synonym(s):
GAD65
About This Item
Recommended Products
biological source
mouse
Quality Level
antibody form
purified immunoglobulin
antibody product type
primary antibodies
clone
GAD-6, monoclonal
species reactivity
human, rat
manufacturer/tradename
Chemicon®
technique(s)
immunohistochemistry: suitable
western blot: suitable
isotype
IgG2a
NCBI accession no.
UniProt accession no.
shipped in
dry ice
target post-translational modification
unmodified
Gene Information
human ... GAD2(2572)
General description
Specificity
Immunogen
Application
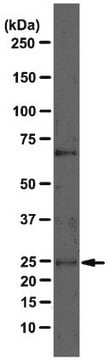
Immunohystochemical Staining Procedures
The following procedure was developed to localize GAD in rat brain sections of cerebellum. Perform all steps at room temperature unless otherwise indicated. Where normal serum is indicated, use normal serum from the same species as the source of the secondary antibody.This procedure represents suggested guidelines for the use of anti-GAD. Fixation regimen, antibody concentrations, and incubation conditions for a given experimental system should be determined empirically.
1. Perfuse rats with 100 mM phosphate buffer, pH 7.4, containing 1% paraformaldehyde, 0.34% L-lysine, and 0.05% sodium m-periodate (1% PLP).
2. Postfix brains in 1% PLP for 1-2 hours. Longer fixation times may reduce labeling intensity.
3. Transfer brains to 100 mM phosphate buffer containing 30% sucrose, and gently agitate on a shaker platform at +4°C for 48-60 hours.
4. Using a sliding microtome, cut 30 mm sections of frozen cerebellum. As the sections are cut, collect them in a vial of cold 100 mM phosphate buffer.
5. Incubate sections in phosphate-buffered saline (PBS) containing 1.5% normal serum and 0.2% TritonX-100 for 30 minutes.
6. On a shaker platform, incubate sections with anti-GAD (diluted in PBS containing 1.5% normal serum and 0.2% Triton X-100 to a final antibody concentration of 1 mg/ml) for 12-36 hours at +4°C.
7. On a shaker platform, rinse sections eight times, 10-15 minutes per rinse, in PBS.
8. Detect with a standard secondary antibody detection system (Hsu et al., 1981; Falini & Taylor, 1983; Harlow & Lane, 1988; Taylor, 1978).
9. Mount sections, dehydrate, and apply coverslips.
Neuroscience
Neurotransmitters & Receptors
Target description
Physical form
Storage and Stability
Analysis Note
Brain tissue
Other Notes
Legal Information
Disclaimer
Not finding the right product?
Try our Product Selector Tool.
recommended
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - Aquatic Chronic 3
Storage Class Code
13 - Non Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Articles
Human iPSC neural differentiation media and protocols used to generate neural stem cells, neurons and glial cell types.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service