All Photos(2)
About This Item
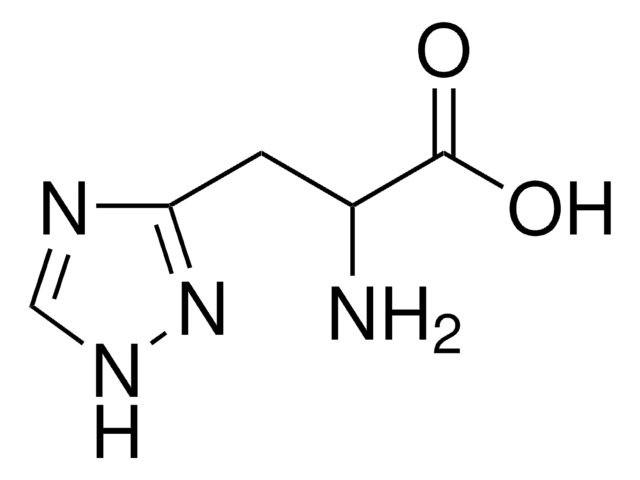
Empirical Formula (Hill Notation):
C10H11N3O2 · H2O
CAS Number:
Molecular Weight:
223.23
MDL number:
UNSPSC Code:
12352209
PubChem Substance ID:
NACRES:
NA.26
Recommended Products
product name
DL-7-Azatryptophan hydrate,
Assay
≥98% (TLC)
form
powder
color
white to off-white
storage temp.
−20°C
SMILES string
O.NC(Cc1c[nH]c2ncccc12)C(O)=O
InChI
1S/C10H11N3O2.H2O/c11-8(10(14)15)4-6-5-13-9-7(6)2-1-3-12-9;/h1-3,5,8H,4,11H2,(H,12,13)(H,14,15);1H2
InChI key
PXDRHYQAIUZKHN-UHFFFAOYSA-N
Biochem/physiol Actions
DL-7-Azatryptophan is a racemic mixture of D- and L-7-azatryptophan which together with L-tryptophan is a synergistic inducer of tryptophan oxygenase of Pseudomonas acidovorans. DL-7-Azatryptophan inhibits photosynthetic carbon assimilation, photosynthetic oxygen evolution and nitrogen metabolism in Anabaena sp. Strain 1F, a marine filamentous, heterocystous cyanobacterium.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
P Cioni et al.
Biochemical and biophysical research communications, 248(2), 347-351 (1998-07-24)
The tryptophan analogues 7-azaindole (7-Aza W) and 5-hydroxytryptophan (5-OH W) have a significant absorbance between 310-320 nm, which allows them to act as selective luminescence probes in protein mixtures containing a large number of tryptophan residues. To assess the potential
C H Chen et al.
Journal of bacteriology, 169(3), 1107-1113 (1987-03-01)
The addition of DL-7-azatryptophan (AZAT), a tryptophan analog, to continuous cultures of Anabaena sp. strain CA grown with 10 mM nitrate as the nitrogen source resulted in the differentiation of heterocysts. Analysis of the intracellular amino acid pools of Anabaena
H Rosenfeld et al.
Journal of bacteriology, 97(2), 697-704 (1969-02-01)
The process of induction of tryptophan oxygenase in Pseudomonas acidovorans is typical of many microbial enzyme induction systems, in that it (i) requires cell multiplication and de novo protein synthesis, (ii) is subject to catabolite repression, (iii) results in the
Fernando Formaggio et al.
Advances in experimental medicine and biology, 527, 731-737 (2004-06-23)
Aal and 7-Atrp, quasi-isosteric with Trp, have been inserted together with a TOAC residue in two 3(10)-helical, model hexapeptides. The interaction of photoexcited AA1 and 7-Atrp with the nitroxide group of TOAC was investigated by time resolved EPR. Both peptides
Lars Merkel et al.
Chembiochem : a European journal of chemical biology, 11(3), 305-314 (2010-01-09)
In vivo expression of colored proteins without post-translational modification or chemical functionalization is highly desired for protein studies and cell biology. Cell-permeable tryptophan analogues, such as azatryptophans, have proved to be almost ideal isosteric substitutes for natural tryptophan in cellular
Chromatograms
application for HPLCOur team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service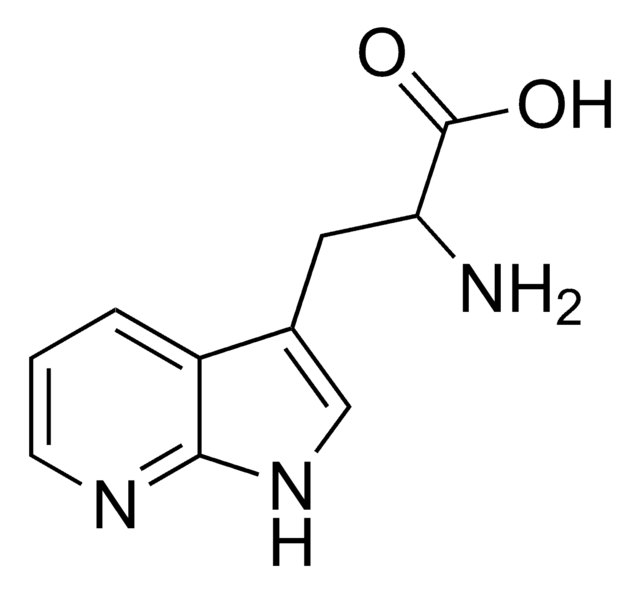



![1,2,3,4-Tetrahydro-9H-pyrido[3,4-b]indole 98%](/deepweb/assets/sigmaaldrich/product/structures/181/460/3d58bc34-1b5c-4295-bbac-3b52085670e8/640/3d58bc34-1b5c-4295-bbac-3b52085670e8.png)