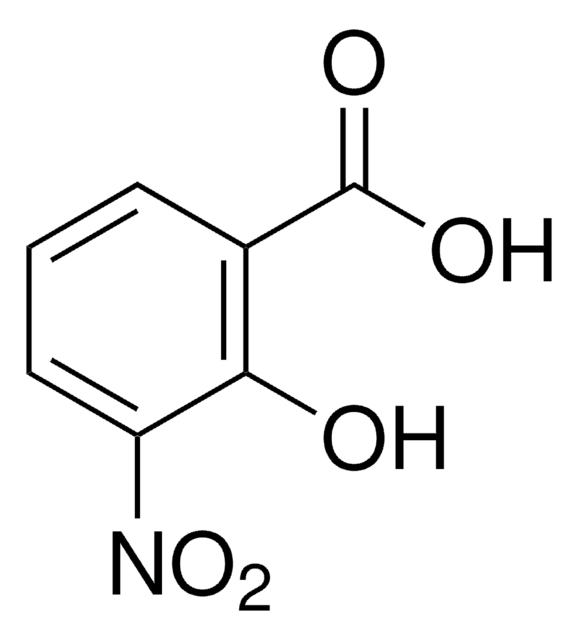
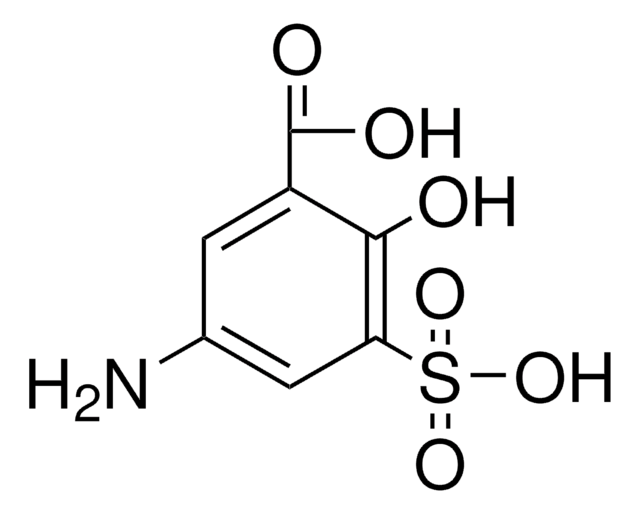
All Photos(4)
About This Item
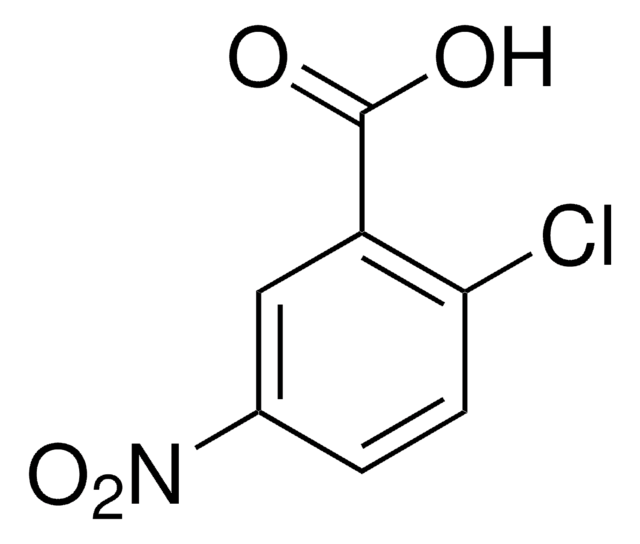
Linear Formula:
ClC6H3(NO2)CO2H
CAS Number:
Molecular Weight:
201.56
Beilstein:
645427
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
Recommended Products
Assay
99%
mp
183-187 °C (lit.)
SMILES string
OC(=O)c1cccc(c1Cl)[N+]([O-])=O
InChI
1S/C7H4ClNO4/c8-6-4(7(10)11)2-1-3-5(6)9(12)13/h1-3H,(H,10,11)
InChI key
JRQDVRIQJJPHEQ-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
2-Chloro-3-nitrobenzoic acid undergoes condensation with aminoquinolines to yield phenylquinolylamines.
Application
2-Chloro-3-nitrobenzoic acid was used as reagent during the synthesis of new biprivileged molecular scaffolds of tetracyclic indolo-benzodiazepines and indolo-quinoxalines. It was used in a microwave-promoted Ulmann condensation with 2-aminopyridines.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Sens. 1
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Synthetic Communications, 36, 1715-1715 (2006)
Indrajeet J Barve et al.
Chemistry, an Asian journal, 7(7), 1684-1690 (2012-04-19)
The present article describes the design and synthesis of new biprivileged molecular scaffolds with diverse structural features. Commercially available, simple heterocyclic building blocks such as 4-fluoro-3-nitrobenzoic acid, 2-chloro-3-nitrobenzoic acid, and indoline were utilized for the synthesis of the novel heterocycles.
J B Bongui et al.
Chemical & pharmaceutical bulletin, 49(9), 1077-1080 (2001-09-18)
Condensation of either 2-bromobenzoic acid (4) or 2-chloro-3-nitrobenzoic acid (5) with suitable aminoquinolines 6-8 afforded phenylquinolylamines 9-13. Acid mediated cyclization gave the corresponding 12H-benzo[b][1,7]phenanthrolin-7-ones 14 and 15, and 12H-benzo[b][1,10]phenanthrolin-7-ones 16-18. Compounds 14, 16, and 17 were subsequently N-methylated to 6-demethoxyacronycine
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service