O0376
Oxalic acid dihydrate
ReagentPlus®, ≥99.0% (GC)
Synonym(s):
Ethanedioic acid dihydrate
About This Item
Recommended Products
vapor density
4.4 (vs air)
Quality Level
vapor pressure
<0.01 mmHg ( 20 °C)
product line
ReagentPlus®
Assay
≥99.0% (GC)
mp
104-106 °C (lit.)
SMILES string
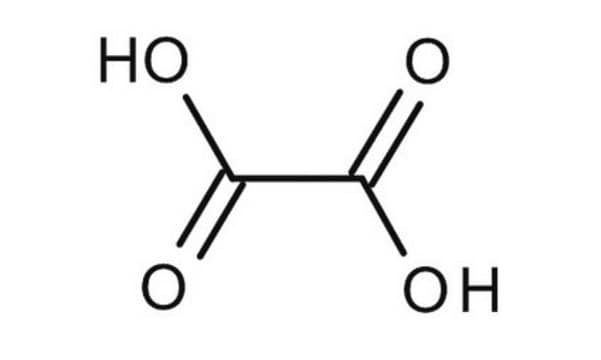
[H]O[H].[H]O[H].OC(=O)C(O)=O
InChI
1S/C2H2O4.2H2O/c3-1(4)2(5)6;;/h(H,3,4)(H,5,6);2*1H2
InChI key
GEVPUGOOGXGPIO-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
- As a catalyst in the preparation of tetrahydroquinoline derivatives via imino Diels-Alder reaction.
- As a homogeneous catalyst in the preparation of highly functionalized piperidines via multi-component reaction.
- As an oxidant for the formation of triazolinediones from corresponding urazoles and bisurazoles via oxidation.
- In the transformation of thiols to nitrosothiols by reacting with sodium nitrite.
Legal Information
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Dermal - Acute Tox. 4 Oral - Eye Dam. 1
Storage Class Code
11 - Combustible Solids
WGK
WGK 1
Personal Protective Equipment
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service