All Photos(1)
About This Item
Empirical Formula (Hill Notation):
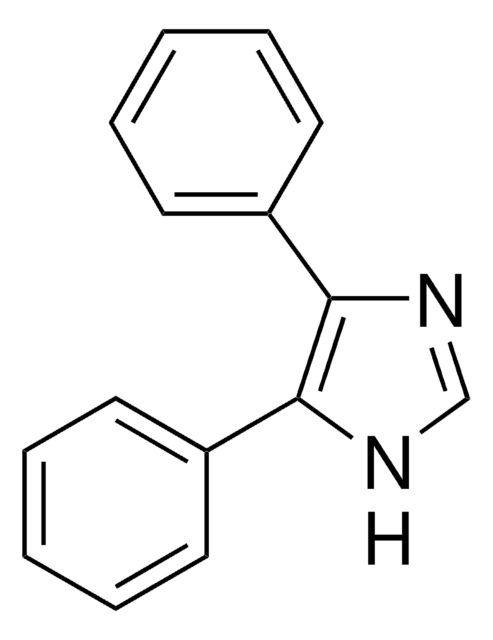
C9H8N2
CAS Number:
Molecular Weight:
144.17
Beilstein:
2969
EC Number:
MDL number:
UNSPSC Code:
12352005
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
97%
mp
128-131 °C (lit.)
solubility
acetone: soluble 25 mg/mL, clear, colorless to yellow (typical)
functional group
phenyl
SMILES string
c1ccc(cc1)-c2c[nH]cn2
InChI
1S/C9H8N2/c1-2-4-8(5-3-1)9-6-10-7-11-9/h1-7H,(H,10,11)
InChI key
XHLKOHSAWQPOFO-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Application
4-Phenylimidazole was used to investigate the protein-ligand interactions in cytochrome P450 from the thermoacidophile Picrophilus torridus. It was used as heme ligand during the crystallization of recombinant human indoleamine 2,3-dioxygenase. It was used in the synthesis of complexes of copper and cobalt.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
J K Yano et al.
The Journal of biological chemistry, 275(40), 31086-31092 (2000-06-22)
The structure of the first P450 identified in Archaea, CYP119 from Sulfolobus solfataricus, has been solved in two different crystal forms that differ by the ligand (imidazole or 4-phenylimidazole) coordinated to the heme iron. A comparison of the two structures
T Kimura et al.
Journal of medicinal chemistry, 36(11), 1641-1653 (1993-05-28)
In our continuing search to find systemically bioavailable ACAT (acyl-CoA:cholesterol O-acyltransferase) inhibitors with more potent antiatherosclerotic effect than N-[2-(dimethylamino)-6-[3-(5-methyl-4-phenyl-1H-imidazol-1-yl)propoxy] phenyl]-N'-pentylurea (3), a series of phenylureas linked to 4-phenylimidazole were synthesized and evaluated for in vitro inhibitory activity toward both aortic
Winny W Ho et al.
Biochemistry, 47(7), 2071-2079 (2008-01-17)
The crystal structure of a cytochrome P450 from the thermoacidophile Picrophilus torridus, CYP231A2 (PTO1399), has been solved. This structure reveals a wide open substrate access channel. To better understand ligand-induced structural transitions in CYP231A2, protein-ligand interactions were investigated using 4-phenylimidazole.
X-RAY STUDIES AND ANTIBACTERIAL ACTIVITY IN COPPER AND COBALT COMPLEXES WITH IMIDAZOLE DERIVATIVE LIGANDS.
ATRIA a, et al.
Journal of the Chilean Chemical Society, 56(3), 786-792 (2011)
M Spatzenegger et al.
Molecular pharmacology, 59(3), 475-484 (2001-02-17)
The molecular basis for reversible inhibition of rabbit CYP2B4 and CYP2B5 and rat CYP2B1 by phenylimidazoles was assessed with active-site mutants and new three-dimensional models based on the crystal structure of CYP2C5. 4-Phenylimidazole was 17- to 32-fold more potent toward
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service