Y0001969
Etanercept
European Pharmacopoeia (EP) Reference Standard
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Productos recomendados
grado
pharmaceutical primary standard
familia API
etanercept
fabricante / nombre comercial
EDQM
aplicaciones
pharmaceutical
Formato
neat
¿Está buscando productos similares? Visita Guía de comparación de productos
Descripción general
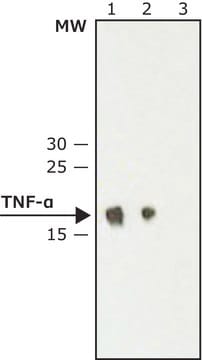
Etanercept is one of the biologic disease-modifying anti-rheumatic drugs (DMARDs) used to treat rheumatoid arthritis (RA). It acts as a tumor necrosis factor (TNF) antagonist by binding with TNF cytokine or protein, thereby blocking its interaction with cell surface receptors. Thus, etanercept effectively relieves joint inflammation from RA and lowers the disease activity.
This product is provided as delivered and specified by the issuing Pharmacopoeia. All information provided in support of this product, including SDS and any product information leaflets have been developed and issued under the Authority of the Issuing Pharmacopoeia. For further information and support please go to the website of the issuing Pharmacopoeia.
This product is provided as delivered and specified by the issuing Pharmacopoeia. All information provided in support of this product, including SDS and any product information leaflets have been developed and issued under the Authority of the Issuing Pharmacopoeia. For further information and support please go to the website of the issuing Pharmacopoeia.
Aplicación
This product is intended for laboratory tests as prescribed in the EP only. Established for use with the monograph(s): 2895.
Envase
The product is delivered as supplied by the issuing Pharmacopoeia. For the current unit quantity, please visit the EDQM reference substance catalogue.
Otras notas
Sales restrictions may apply.
Producto relacionado
Referencia del producto
Descripción
Precios
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Elija entre una de las versiones más recientes:
Certificados de análisis (COA)
Lot/Batch Number
It looks like we've run into a problem, but you can still download Certificates of Analysis from our Documentos section.
Si necesita más asistencia, póngase en contacto con Atención al cliente
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Development of a sensitive enzyme-linked immunosorbent assay for the measurement of biologically active etanercept in patients with ankylosing spondylitis
Wang L, et al.
Journal of Chromatography. B, Biomedical Applications, 1008, 219-224 (2016)
Etanercept
European Pharmacopoeia Commission and Directorate for the Quality of Medicines & Healthcare
European pharmacopoeia, 4979-4983 (2021)
Etanercept concentration in patients with rheumatoid arthritis and its potential influence on treatment decisions: a pilot study
Daien CI, et al.
The Journal of Rheumatology, 39(8), 1533-1538 (2012)
Characterization and application of a unique panel of monoclonal antibodies generated against etanercept
Detrez I, et al.
Journal of Immunology, 196(6), 2879-2884 (2016)
Rheumatiod Arthritis: An Updated Overview of Latest Therapy and Drug Delivery
Kesharwani D, et al.
Journal of pharmacopuncture, 22(4), 210-224 (2019)
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico