I17451
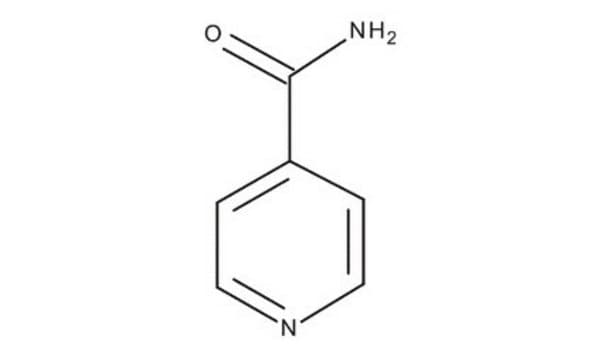
Isonicotinamide
ReagentPlus®, 99%
Sinónimos:
Pyridine-4-carboxylic acid amide
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula empírica (notación de Hill):
C6H6N2O
Número de CAS:
Peso molecular:
122.12
Beilstein:
2173
Número CE:
Número MDL:
Código UNSPSC:
12352100
ID de la sustancia en PubChem:
NACRES:
NA.22
Productos recomendados
Línea del producto
ReagentPlus®
Ensayo
99%
mp
155-157 °C (lit.)
cadena SMILES
NC(=O)c1ccncc1
InChI
1S/C6H6N2O/c7-6(9)5-1-3-8-4-2-5/h1-4H,(H2,7,9)
Clave InChI
VFQXVTODMYMSMJ-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Categorías relacionadas
Aplicación
Isonicotinamide (pyridine-4-carboxamide) can be used as a heterocyclic building block to synthesize:
It can also be used as a co-former with active pharmaceutical ingredients (APIs) to prepare co-crystals.
- 4-oxo-1,3-thiazinan-3-yl isonicotinamide derivatives as potential anti-tubercular agents.
- Organotin(IV) complexes of isonicotinamide via synthesis of phosphoramidate ligands for various biological activity studies.
- Bis-pyridinium isonicotinamide derivatives of 2-(hydroxyimino)-N-(pyridin-3-yl)acetamide as potent reactivators sarin.
It can also be used as a co-former with active pharmaceutical ingredients (APIs) to prepare co-crystals.
Información legal
ReagentPlus is a registered trademark of Merck KGaA, Darmstadt, Germany
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Equipo de protección personal
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
QSAR, docking studies of 1, 3-thiazinan-3-yl isonicotinamide derivatives for antitubercular activity
Chitre TS, et al.
Computational Biology and Chemistry, 68, 211-218 (2017)
New organotin (IV) complexes of nicotinamide, isonicotinamide and some of their novel phosphoric triamide derivatives: Syntheses, spectroscopic study and crystal structures
Gholivand K, et al.
Journal of Organometallic Chemistry, 695(9), 1383-1391 (2010)
Shaun R Stauffer et al.
Bioorganic & medicinal chemistry letters, 17(6), 1788-1792 (2007-01-30)
A series of low-molecular weight 2,6-diamino-isonicotinamide BACE-1 inhibitors containing an amine transition-state isostere were synthesized and shown to be highly potent in both enzymatic and cell-based assays. These inhibitors contain a trans-S,S-methyl cyclopropane P(3) which bind BACE-1 in a 10s-loop
Gareth Arnott et al.
Organic letters, 10(14), 3089-3092 (2008-06-17)
Treatment of N-arylisonicotinamides with trifluoromethanesulfonic anhydride triggers intramolecular nucleophilic attack of the aryl ring on the 4-position of the pyridinium intermediate. The products are spirocyclic dihydropyridines which can be converted to valuable spirocyclic piperidines related to biologically active molecules such
Sarah Boyd et al.
Journal of pharmaceutical sciences, 99(9), 3779-3786 (2010-07-29)
The solubility and crystal growth of the 1:1 cocrystal between benzoic acid and isonicotinamide from 95% ethanol was studied through the creation of a ternary phase diagram at differing temperatures and turbidity measurements. From the solubility measurements thermodynamic properties of
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico