81900
Propionitrile
purum, ≥99.0% (GC)
Synonym(s):
PPN, Ethyl cyanide
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
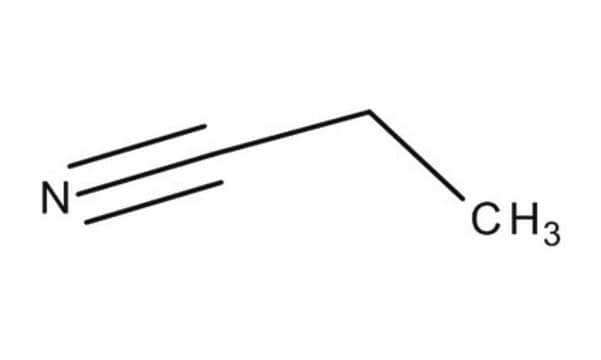
Linear Formula:
CH3CH2CN
CAS Number:
Molecular Weight:
55.08
Beilstein:
773680
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
grade
purum
Quality Level
Assay
≥99.0% (GC)
form
liquid
refractive index
n20/D 1.366 (lit.)
n20/D 1.366
bp
97 °C (lit.)
mp
−93 °C (lit.)
density
0.772 g/mL at 25 °C (lit.)
functional group
nitrile
SMILES string
CCC#N
InChI
1S/C3H5N/c1-2-3-4/h2H2,1H3
InChI key
FVSKHRXBFJPNKK-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Application
- Propionitrile (PPN) is an effective solvent for catalytic asymmetric aldol reaction of a silyl enol ether with aldehydes in the presence of a chiral tin(II) Lewis acid catalyst.
- The co-solvent formed by mixing PPN with acetonitrile can be used to fabricate polymer gel electrolytes (PGEs) of dye-sensitized solar cells (DSSCs), which lead to enhanced stability of gel-state DSSCs.
- PPN can be used as a solvent for the Brønsted acid-catalyzed synthesis of N-alkyl cis-aziridines via [2+1] annulation of a diazo compound formed by the combination of an acetate and enolate. The process does not involve the use of metals or reagents and only atomic nitrogen as a co-product.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 2 Dermal - Acute Tox. 2 Oral - Acute Tox. 4 Inhalation - Eye Irrit. 2 - Flam. Liq. 2
Storage Class Code
3 - Flammable liquids
WGK
WGK 1
Flash Point(F)
42.8 °F - closed cup
Flash Point(C)
6 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Stability improvement of gel-state dye-sensitized solar cells by utilization the co-solvent effect of propionitrile/acetonitrile and 3-methoxypropionitrile/acetonitrile with poly (acrylonitrile-co-vinyl acetate).
Venkatesan S, et al.
Journal of Power Sources, 274, 506-511 (2015)
The Br?nsted acid-catalyzed direct Aza-Darzens synthesis of N-alkyl cis-aziridines.
Williams A L and Johnston J N
Journal of the American Chemical Society, 126(6), 1612-1613 (2004)
Catalytic asymmetric aldol-type reaction using a chiral tin (II) Lewis acid.
Kobayashi S, et al.
Tetrahedron, 49(9), 1761-1772 (1993)
Sung Hee Joo et al.
Environmental science & technology, 41(4), 1288-1296 (2007-06-28)
The decreasing availability of pristine water supplies is prompting drinking water utilities to exploit waters impacted by wastewater effluents and agricultural runoff. As these waters feature elevated organic nitrogen concentrations, the pathways responsible for transformation of organic nitrogen into toxic
D Somjen et al.
Journal of cellular biochemistry, 112(2), 625-632 (2011-01-27)
In cultured human osteoblasts estradiol-17β (E2) modulated DNA synthesis, the specific activity of creatine kinase BB (CK), 12 and 15 lipoxygenase (LO) mRNA expression and formation of 12- and 15-hydroxyeicosatetraenoic acid (HETE). We now investigate the response of human bone
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service