523976
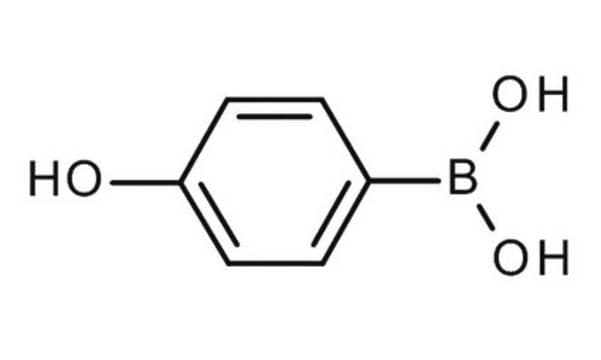
4-Hydroxyphenylboronic acid
≥95.0%
Sinónimos:
(p-Hydroxyphenyl)boronic acid, 4-Hydroxybenzeneboronic acid, p-hydroxy-benzeneboronic acid
About This Item
Productos recomendados
assay
≥95.0%
form
solid
mp
>230 °C (lit.)
SMILES string
OB(O)c1ccc(O)cc1
InChI
1S/C6H7BO3/c8-6-3-1-5(2-4-6)7(9)10/h1-4,8-10H
InChI key
COIQUVGFTILYGA-UHFFFAOYSA-N
Categorías relacionadas
Application
- Suzuki-Miyaura coupling and Stille coupling reactions.
- Palladium-catalyzed aminocarbonylation and cross-coupling reactions.
- Suzuki reaction for preparation of bio-supported palladium nanoparticles as phosphine-free catalysts.
- Cu2O-catalyzed aerobic oxidative cross-coupling of tetrazoles.
It can also be used to prepare/promote:
- PDK1 inhibitory activity (cancer cell growth, survival, and tumorigenesis inhibitor).
- Rod-like dendronized polymers containing G4 and G5 ester dendrons via macromonomer approach by living ROMP.
- Estrone-derived cyclopamine analogs as Sonic Hedgehog signaling inhibitors for anti-cancer chemotherapeutics.
- Enzymatic inhibitors for the treatment of Gram-negative bacterial infections.
- Oligoarenes by Suzuki-Miyaura palladium-catalyzed cross-coupling.
Other Notes
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico