905313
Rh2(S-TCPTAD)4
Synonym(s):
Davies dirhodium catalyst
Sign Into View Organizational & Contract Pricing
All Photos(3)
About This Item
Empirical Formula (Hill Notation):
C80H64Cl16N4O16Rh2
CAS Number:
Molecular Weight:
2110.44
UNSPSC Code:
12352101
NACRES:
NA.22
Recommended Products
form
powder or crystals
Application
This dirhodium catalyst developed by the Davies lab can form C-C bonds at the most accessible tertiary C-H position with control of both regioselectivity and absolute configuration.
Other Notes
Formation of Tertiary Alcohols from the Rhodium-Catalyzed Reactions of Donor/Acceptor Carbenes with Esters
Harnessing the β-Silicon Effect for Regioselective and Stereoselective Rhodium(II)-Catalyzed C-H Functionalization by Donor/Acceptor Carbenes Derived from 1-Sulfonyl-1,2,3-triazoles
Site-selective and stereoselective functionalization of non-activated tertiary C-H bonds
Harnessing the β-Silicon Effect for Regioselective and Stereoselective Rhodium(II)-Catalyzed C-H Functionalization by Donor/Acceptor Carbenes Derived from 1-Sulfonyl-1,2,3-triazoles
Site-selective and stereoselective functionalization of non-activated tertiary C-H bonds
related product
Product No.
Description
Pricing
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Atsushi D Yamaguchi et al.
Journal of the American Chemical Society, 137(2), 644-647 (2015-01-07)
Syntheses of dictyodendrins A and F have been achieved using a sequential C-H functionalization strategy. The N-alkylpyrrole core is fully functionalized by means of a rhodium(I)-catalyzed C-H arylation at the C3-position, a rhodium(II)-catalyzed double C-H insertion at the C2- and
Shigeki Sato et al.
Chemical communications (Cambridge, England), (41), 6264-6266 (2009-10-15)
A versatile, highly enantiocontrolled entry to the spiro-beta-lactam core of chartellines has been developed by expanding the scope of oxidative nitrogen atom transfer methodology based on chiral Rh-nitrenoid species.
Enantioselective intramolecular aza-spiroannulation onto benzofurans using chiral rhodium catalysis.
Shibuta T, et al.
Heterocycles, 89, 631-639 (2014)
Liangbing Fu et al.
Organic letters, 16(11), 3036-3039 (2014-05-21)
A rhodium-catalyzed asymmetric synthesis of β-lactones via intramolecular C-H insertion into the ester group of aryldiazoacetates has been developed. The β-lactones were synthesized in high yields and with high levels of diastereo- and enantioselectivity. Halo and trifluoromethyl substituents at the
Ravisekhara P Reddy et al.
Organic letters, 8(22), 5013-5016 (2006-10-20)
The dirhodium tetracarboxylate, Rh(2)(S-TCPTAD)(4), derived from adamantylglycine, is an effective chiral catalyst for both inter- and intramolecular C-H aminations. [reaction: see text]
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service
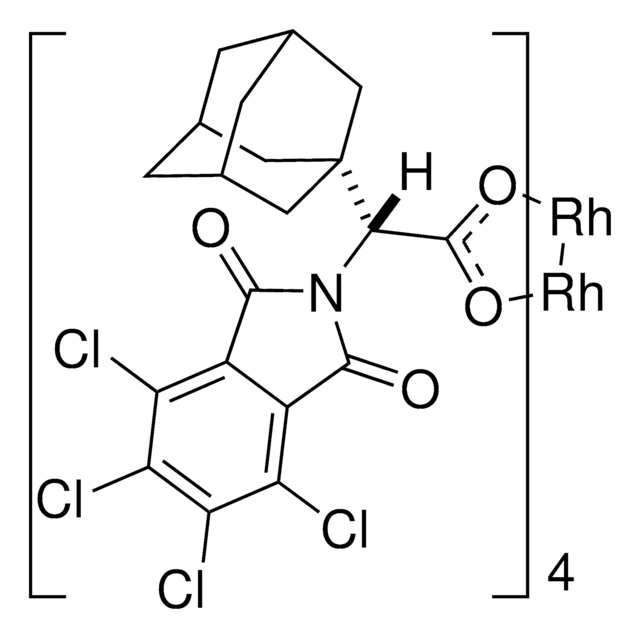




![Bis[rhodium(α,α,α′,α′-tetramethyl-1,3-benzenedipropionic acid)] 95%](/deepweb/assets/sigmaaldrich/product/structures/102/178/d1171a49-0358-406b-8b32-04324dbf9c02/640/d1171a49-0358-406b-8b32-04324dbf9c02.png)