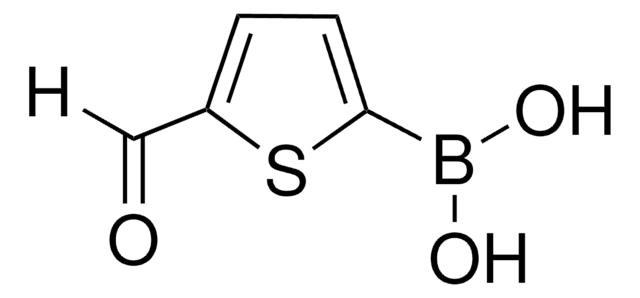
900555
(N-Isocyanoimino)triphenylphosphorane
95%
Synonym(s):
Pinc
About This Item
Recommended Products
Quality Level
Assay
95%
form
solid
application(s)
peptide synthesis
InChI
1S/C19H15N2P/c1-20-21-22(17-11-5-2-6-12-17,18-13-7-3-8-14-18)19-15-9-4-10-16-19/h2-16H
InChI key
NIDTXBFHPXMXTR-UHFFFAOYSA-N
General description

Application
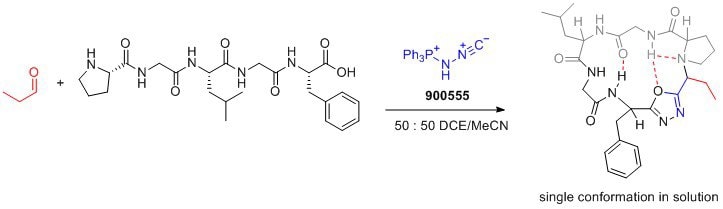
Features and Benefits
- Bench-stable solid: Pinc is a stable reagent that can be easily handled and stored.
- Facilitates cyclization and incorporation of conformational control element: Pinc enables the formation of peptide macrocycles with a desired conformation, leading to improved drug properties.
- Amphoteric properties: Pinc′s amphoteric nature allows for the design of new multicomponent reactions, expanding its synthetic capabilities.
- Desired drug properties: The resulting peptide macrocycles exhibit enhanced membrane permeability, lipophilicity, and aqueous solubility, making them desirable for drug development.
- Versatile transformations: Pinc′s ability to form oxadiazoles opens up opportunities for the development of novel synthetic transformations.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Articles
Isocyanides are widely used reagents in organic synthesis, with applications ranging from materials science to drug discovery.
Related Content
The Yudin laboratory is known for the development of amphoteric molecules and their application in synthesis. The corresponding reagents possess nucleophilic and electrophilic functional groups that do not prematurely react with each other.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service
![Thieno[3,2-b]thiophene-2,5-dicarboxaldehyde 96%](/deepweb/assets/sigmaaldrich/product/structures/137/771/57dfbc98-f02d-4773-bc11-3e8b861ad74b/640/57dfbc98-f02d-4773-bc11-3e8b861ad74b.png)