All Photos(1)
About This Item
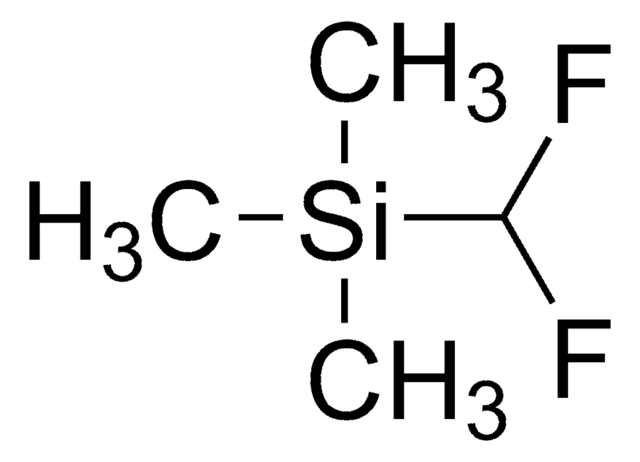
Linear Formula:
(CH3)2C=CHOSi(CH3)3
CAS Number:
Molecular Weight:
144.29
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
Recommended Products
Assay
99%
form
liquid
refractive index
n20/D 1.409 (lit.)
bp
114-116 °C/650 mmHg (lit.)
density
0.785 g/mL at 25 °C (lit.)
SMILES string
C\C(C)=C\O[Si](C)(C)C
InChI
1S/C7H16OSi/c1-7(2)6-8-9(3,4)5/h6H,1-5H3
InChI key
KZVPFSJPLBOVLO-UHFFFAOYSA-N
General description
2-Methyl-1-(trimethylsilyloxy)-1-propene is an enol ether.
Application
2-Methyl-1-(trimethylsilyloxy)-1-propene may be used as reagent in the direct conversion of α-amino acids into β-amino aldehydes.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Flam. Liq. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
3 - Flammable liquids
WGK
WGK 3
Flash Point(F)
57.2 °F - closed cup
Flash Point(C)
14 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
L Nassi et al.
Mutation research, 198(1), 53-60 (1988-03-01)
Generation of triplet ketones, either chemically through thermal decomposition of 3-hydroxymethyl-3,4,4-trimethyl-1,2-dioxetane and 3-[N-(pyridino)carbamoyl]methyl-3,4,4-trimethyl-1,2-dioxetane++ + or enzymatically via the aerobic oxidation of isobutyraldehyde trimethylsilyl enol ether catalyzed by horse-radish peroxidase, triggers the SOS function sfiA in E. coli. Although the observed
Carlos J Saavedra et al.
Organic & biomolecular chemistry, 10(22), 4448-4461 (2012-05-05)
A direct method for the transformation of α-amino acids into β-amino aldehydes was developed, and applied to the modification of the C-terminal residue of peptides. The method takes place in good yields and under mild conditions. The application of this
G Cilento et al.
Toxicology letters, 67(1-3), 17-28 (1993-04-01)
Selected enzymatic systems can efficiently produce a product in the electronically excited triplet state. Earlier, only the formation of electronically excited singlet species was known. The formation of triplet species has been demonstrated with both normal substrates/metabolites and with xenobiotics
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service




![[Bis(trifluoroacetoxy)iodo]benzene 97%](/deepweb/assets/sigmaaldrich/product/structures/238/293/71fcde9a-4afb-4cf5-9c22-8d8d68bf1ba4/640/71fcde9a-4afb-4cf5-9c22-8d8d68bf1ba4.png)