530190
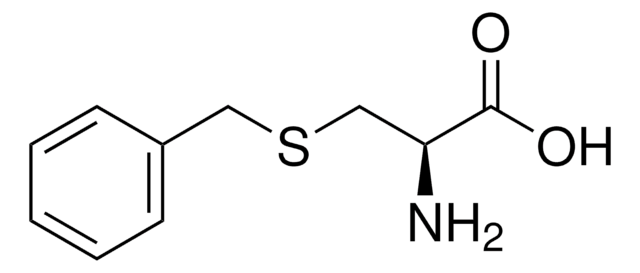
S-Phenyl-L-cysteine
97%
Synonym(s):
3-(Phenylthio)-L-Alanine, 4-Thia-L-homophenylalanine
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
C6H5SCH2CH(NH2)CO2H
CAS Number:
Molecular Weight:
197.25
Beilstein:
2268203
MDL number:
UNSPSC Code:
12352209
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
97%
optical activity
[α]20/D +10°, c = 1.5 in 1 M NaOH
reaction suitability
reaction type: solution phase peptide synthesis
mp
200 °C (dec.) (lit.)
application(s)
peptide synthesis
SMILES string
N[C@@H](CSc1ccccc1)C(O)=O
InChI
1S/C9H11NO2S/c10-8(9(11)12)6-13-7-4-2-1-3-5-7/h1-5,8H,6,10H2,(H,11,12)/t8-/m0/s1
InChI key
XYUBQWNJDIAEES-QMMMGPOBSA-N
Looking for similar products? Visit Product Comparison Guide
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
W E Bechtold et al.
Journal of toxicology and environmental health, 40(2-3), 377-386 (1993-10-01)
Three biomarkers for benzene exposure were developed. The first biomarker, muconic acid in urine, results from the ring opening of a benzene metabolite. A gas chromatography/mass spectroscopy (GC/MS) assay was developed to measure urinary muconic acid, and the analyte in
A A Melikian et al.
Cancer epidemiology, biomarkers & prevention : a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology, 1(4), 307-313 (1992-05-01)
The present study was aimed at the characterization of the major adducts formed by reaction of the metabolites of [14C]benzene with rat hemoglobin in vivo. Groups of 12-week-old male Fisher rats received i.p. injections of a single dose of 10
W E Bechtold et al.
Environmental health perspectives, 104 Suppl 6, 1147-1149 (1996-12-01)
Biological markers of internal dose are useful for improving the extrapolation of health effects from exposures to high levels of toxic air pollutants in animals to low, ambient exposures in humans. Previous results from our laboratory have shown that benzene
W E Bechtold et al.
Carcinogenesis, 13(7), 1217-1220 (1992-07-01)
Results of experiments in our laboratory have shown that benzene is metabolized by animals in part to an intermediate that binds to cysteine groups in hemoglobin to form the adduct S-phenylcysteine (SPC). These results suggested that SPC in hemoglobin may
W E Bechtold et al.
Archives of toxicology, 66(5), 303-309 (1992-01-01)
Benzene is metabolized to intermediates that bind to hemoglobin, forming adducts. These hemoglobin adducts may be usable as biomarkers of exposure. In this paper, we describe the development of a gas chromatography/mass spectroscopy assay for quantitating the binding of the
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service