The product is packaged in bottomless glass bottles with an inserted fused cone. The micro-vials contain the product within an inner cone, while the bottomless outer glass cylinder, serves as a stand. This design is intentional and not a defect.
Select a Size
| Pack Size | SKU | Availability | Price |
|---|
About This Item
Product Name
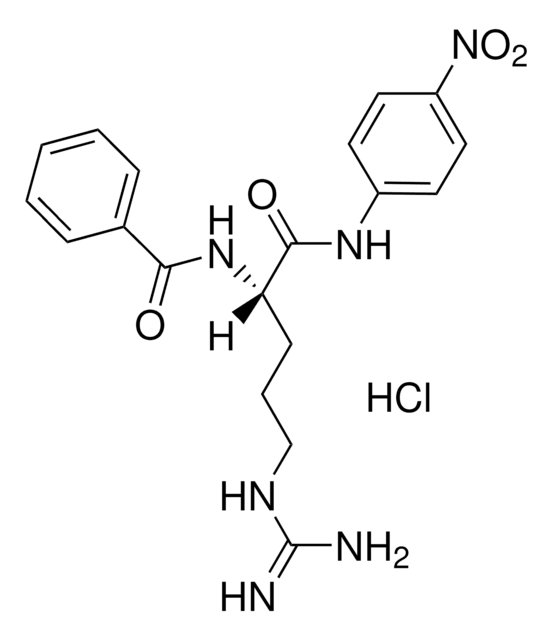
Nα-Benzoyl-DL-arginine 4-nitroanilide hydrochloride, ≥98%
Quality Segment
assay
≥98%
form
powder
color
, white to light yellow to light brown
solubility
DMSO: 50 mg/mL (colorless to dark green-yellow to dark brown-yellow)
storage temp.
−20°C
SMILES string
Cl[H].NC(=N)NCCCC(NC(=O)c1ccccc1)C(=O)Nc2ccc(cc2)[N+]([O-])=O
InChI
1S/C19H22N6O4.ClH/c20-19(21)22-12-4-7-16(24-17(26)13-5-2-1-3-6-13)18(27)23-14-8-10-15(11-9-14)25(28)29;/h1-3,5-6,8-11,16H,4,7,12H2,(H,23,27)(H,24,26)(H4,20,21,22);1H
InChI key
DEOKFPFLXFNAON-UHFFFAOYSA-N
General description
Application
Packaging
1 of 1
This Item | |||
|---|---|---|---|
| assay ≥98% | assay ≥98% (HPLC) | assay ≥98% (TLC) | assay ≥98% (TLC) |
| solubility DMSO: 50 mg/mL (colorless to dark green-yellow to dark brown-yellow) | solubility acetone: water (1:1): 40.00-51.00 mg/mL, clear, colorless to yellow | solubility DMF: 50 mg/mL, clear, colorless to faintly yellow | solubility methanol: 20 mg/mL, clear, colorless to light yellow |
| Quality Level 200 | Quality Level 200 | Quality Level 200 | Quality Level 200 |
| form powder | form powder | form powder | form powder |
| storage temp. −20°C | storage temp. −20°C | storage temp. 2-8°C | storage temp. −20°C |
| color , white to light yellow to light brown | color - | color - | color - |
Storage Class
11 - Combustible Solids
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Global Trade Item Number
| SKU | GTIN |
|---|---|
| B4875-1G | 04061835504572 |
| B4875-500MG | 04061835553839 |
| B4875-100MG | 04061835540709 |
| B4875-10G | 04061835553822 |
| B4875-5G | 04061835504589 |
-
Why do the bottoms of the vials/bottles of product B4875-100MG (Nα-Benzoyl-DL-arginine 4-nitroanilide hydrochloride) appear to have no bottom? How is the product contained within the vials despite this unusual appearance? Is this a packaging defect?
1 answer-
Helpful?
-