A1879
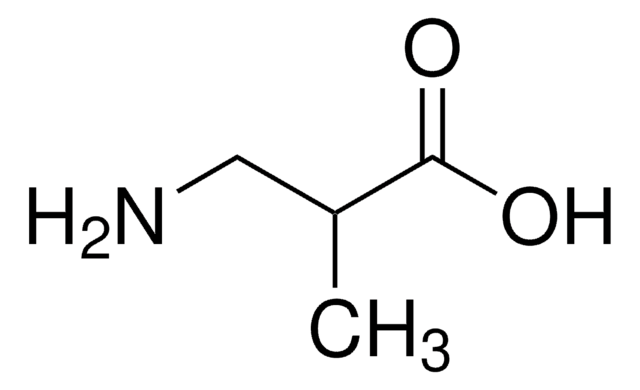
L-2-Aminobutyric acid
≥99% (titration), suitable for ligand binding assays
Synonym(s):
L-α-Aminobutyric acid
About This Item
Recommended Products
product name
L-2-Aminobutyric acid, ≥99% (titration)
Assay
≥99% (titration)
form
powder
technique(s)
ligand binding assay: suitable
color
white
application(s)
peptide synthesis
SMILES string
CC[C@H](N)C(O)=O
InChI
1S/C4H9NO2/c1-2-3(5)4(6)7/h3H,2,5H2,1H3,(H,6,7)/t3-/m0/s1
InChI key
QWCKQJZIFLGMSD-VKHMYHEASA-N
Looking for similar products? Visit Product Comparison Guide
Application
- Targeted metabolomics combined with machine learning to identify and validate new biomarkers for early SLE diagnosis and disease activity.: This study utilizes L-2-aminobutyric acid in targeted metabolomics, enhancing the predictive accuracy for Systemic Lupus Erythematosus (SLE), a crucial development in autoimmune disease diagnostics (Liang J et al., 2024).
- Alcoholamine enhanced fractionation of cellulose from lignocellulosic biomass in ionic liquids.: This research highlights the use of L-2-aminobutyric acid in the process of biomass conversion, offering insights into more efficient biofuel production techniques (Zhu Y et al., 2023).
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Skin Sens. 1
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service