H43806
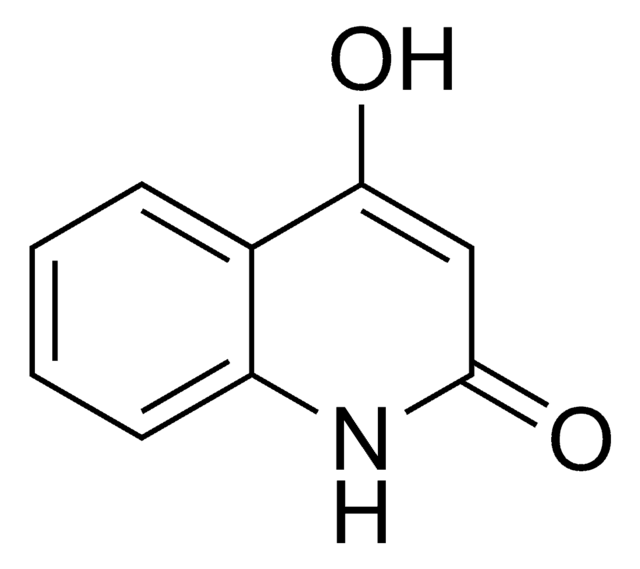
4-Hydroxy-2-methylquinoline
98.5%
Synonym(s):
2-Methyl-4-quinolinol
Sign Into View Organizational & Contract Pricing
All Photos(3)
About This Item
Empirical Formula (Hill Notation):
C10H9NO
CAS Number:
Molecular Weight:
159.18
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
98.5%
SMILES string
Cc1cc(O)c2ccccc2n1
InChI
1S/C10H9NO/c1-7-6-10(12)8-4-2-3-5-9(8)11-7/h2-6H,1H3,(H,11,12)
InChI key
NWINIEGDLHHNLH-UHFFFAOYSA-N
Application
4-Hydroxy-2-methylquinoline can be used as an intermediate in the synthesis of a wide range of medicinally important compounds such as:
- Synthesis of 2-(quinolin-4-yloxy)acetamides as potent antitubercular agents.
- Synthesis of 2-arylethenylquinoline derivatives for the treatment of Alzheimer′s disease.
- Synthesis of 1,10-diethoxy-1H-pyrano[4,3-b]quinolones as antibacterial agents.
- Synthesis of phenylimidazole-pyrazolo[1,5-c]quinazolines as potent phophodiesterase 10A (PDE10A) inhibitors.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Design, synthesis, and biological evaluation of 2-arylethenylquinoline derivatives as multifunctional agents for the treatment of Alzheimer's disease.
Wang X Q, et al.
European Journal of Medicinal Chemistry, 89, 349-361 (2015)
2-(Quinolin-4-yloxy) acetamides are active against drug-susceptible and drug-resistant Mycobacterium tuberculosis strains.
Pissinate K, et al.
ACS Medicinal Chemistry Letters, 7(3), 235-239 (2016)
Synthesis of new 1, 10-diethoxy-1H-pyrano [4, 3-b] quinolines and their antibacterial studies.
Dhanabal T, et al.
Indian J. Chem. B, 45B(02) (2006)
Synthesis and SAR study of new phenylimidazole-pyrazolo [1, 5-c] quinazolines as potent phosphodiesterase 10A inhibitors.
Asproni B, et al.
Bioorganic & Medicinal Chemistry, 19(1), 642-649 (2011)
Quinaldine derivatives: Preparation and biological activity.
Jampilek J, et al.
Medicinal Chemistry, 1(6), 591-599 (2005)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service