144169
2,3-Hexanedione
technical grade, 90%
Synonym(s):
Acetyl butyryl
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Linear Formula:
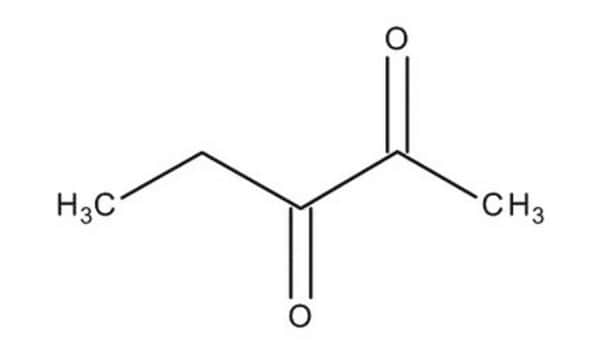
CH3CH2CH2COCOCH3
CAS Number:
Molecular Weight:
114.14
Beilstein:
1699896
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
grade
technical grade
vapor density
3.9 (vs air)
vapor pressure
10 mmHg ( 20 °C)
Assay
90%
form
liquid
refractive index
n20/D 1.412 (lit.)
bp
128 °C (lit.)
density
0.934 g/mL at 25 °C (lit.)
functional group
ketone
SMILES string
CCCC(=O)C(C)=O
InChI
1S/C6H10O2/c1-3-4-6(8)5(2)7/h3-4H2,1-2H3
InChI key
MWVFCEVNXHTDNF-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
2,3-Hexanedione is a major component of the male-produced aggregation pheromone of Anelaphus inflaticollis Chemsak. It reacts with ethylenediamine to yield macrocyclic tetradentate 12-membered nitrogen donor (N4) ligand.
Application
2,3-Hexanedione was used to evaluate the influence of xanthan concentration on the release of aroma compounds in xanthan-thickened food model systems.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Flam. Liq. 3 - STOT RE 2 Inhalation
Target Organs
Respiratory system
Storage Class Code
3 - Flammable liquids
WGK
WGK 2
Flash Point(F)
82.4 °F - closed cup
Flash Point(C)
28 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
A M Ray et al.
Environmental entomology, 38(5), 1462-1466 (2009-10-15)
We report the identification and field bioassays of a major component of the male-produced aggregation pheromone of Anelaphus inflaticollis Chemsak, an uncommon desert cerambycine beetle. Male A. inflaticollis produced a sex-specific blend of components that included (R)-3-hydroxyhexan-2-one, (S)-2-hydroxyhexan-3-one, 2,3-hexanedione, and
Sulekh Chandra et al.
Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy, 60(13), 3079-3085 (2004-10-13)
Complexes of Cr(III), Co(II), Ni(II) and Cu(II) containing a novel macrocyclic tetradentate nitrogen donor (N4) ligand prepared via reaction of 2,3-hexanedione and ethylenediamine has been prepared and characterized. The newly synthesized ligand (L) and its complexes have been characterized on
Emerson S Lacey et al.
Journal of chemical ecology, 30(8), 1493-1507 (2004-11-13)
This is the first fully verified report of an aggregation pheromone produced by a cerambycid beetle species. Field bioassays with adult Neoclytus acuminatus acuminatus (F.) (Coleoptera: Cerambycidae) revealed that males produce a pheromone that attracts both sexes. Extracts of odors
Egle Bylaite et al.
Journal of agricultural and food chemistry, 53(9), 3577-3583 (2005-04-28)
The influence of xanthan concentration (0, 0.02, 0.1, 0.4, and 0.8% w/w) and bulk viscosity on the release of 20 aroma compounds of different chemical classes (5 aldehydes, 4 esters, 5 ketones, 3 alcohols, and 3 terpenes) was evaluated in
Molecular mechanism of hexane neuropathy: significant differences in pharmacokinetics between 2.3-, 2.4-, and 2.5-hexanedione.
K Iwasaki et al.
Industrial health, 22(3), 177-187 (1984-01-01)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service