208743
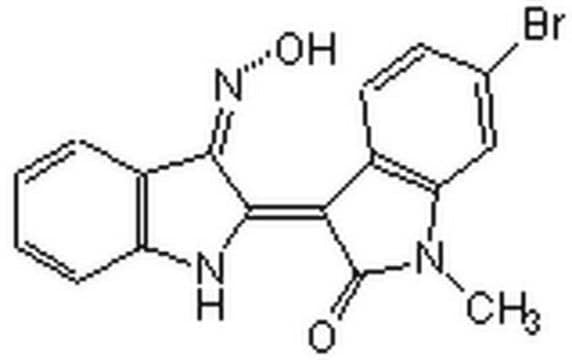
Calpain Inhibitor XI
The Calpain Inhibitor XI, also referenced under CAS 145731-49-3, controls the biological activity of Calpain. This small molecule/inhibitor is primarily used for Protease Inhibitors applications.
Synonym(s):
Calpain Inhibitor XI, Z-L-Abu-CONH(CH₂)₃-morpholine
About This Item
Recommended Products
Quality Level
Assay
≥95% (HPLC)
form
solid
manufacturer/tradename
Calbiochem®
storage condition
OK to freeze
desiccated (hygroscopic)
color
white to off-white
solubility
DMSO: 5 mg/mL
shipped in
ambient
storage temp.
2-8°C
General description
Biochem/physiol Actions
calpain 1, calpain 2
Packaging
Warning
Sequence
Physical form
Reconstitution
Other Notes
DeBiasi, R.L., et al. 2001. J. Virol.75, 351.
Saatman, K.E., et al. 2000. J. Cereb. Blood Flow Metab.20, 66.
Stelmasiak, Z., et al. 2000. Med. Sci. Monit.6, 426.
Blomgren, K., et al. 1999. J. Biol. Chem.274, 14046.
James, T., et al. 1998. J. Neurosci. Res.51, 218.
Li, Z., et al. 1996. J. Med. Chem.39, 4089.
Saatman, K.E, et al. 1996. Proc. Natl. Acad. Sci. USA93, 3428.
Bartus, R.T., et al. 1995. Neurol. Res.17, 249.
Bartus, R.T., et al. 1994. Stroke25, 2265.
Legal Information
Storage Class Code
11 - Combustible Solids
WGK
WGK 1
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Related Content
Select different protease inhibitor types based on your needs to prevent protein degradation during isolation and characterization and safeguard proteins in sample prep.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service