H31859
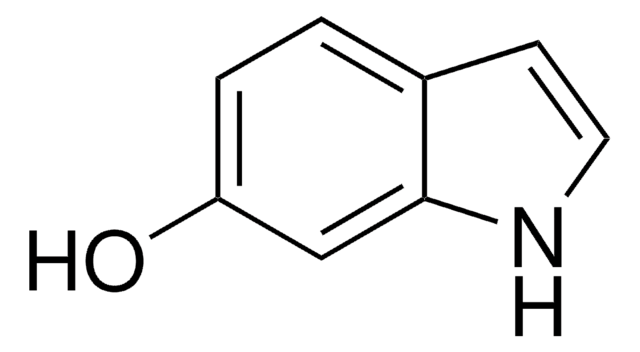
5-Hydroxyindole
97%
Synonym(s):
5-Indolol, 5-Hydroxyindole, NSC 87503
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C8H7NO
CAS Number:
Molecular Weight:
133.15
Beilstein:
112349
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
97%
mp
106-108 °C (lit.)
SMILES string
Oc1ccc2[nH]ccc2c1
InChI
1S/C8H7NO/c10-7-1-2-8-6(5-7)3-4-9-8/h1-5,9-10H
InChI key
LMIQERWZRIFWNZ-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Application
- Reactant for preparation of (oxoimidazolidinyl/oxopyrimidinyl)benzenesulfonates as antitumor agents and tubulin inhibitors
- Reactant for preparation of anthranilic acids
- Reactant for preparation of indole compounds as dopamine D2 receptor antagonists
- Reactant for preparation of naphthalimide- or carbazole-containing human β-adrenoceptor ligands
- Reactant for preparation of melanins as nature-inspired radioprotectors
- Reactant for preparation of 5-vinyl-3-pyridinecarbonitriles as PKCθ inhibitors
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
David Robinson et al.
The journal of physical chemistry. B, 113(8), 2535-2541 (2009-02-07)
We have investigated the absorption and emission spectrum of 5-hydroxyindole in the gas phase and in various solvents. 5-Hydroxyindole is the fluorophore of the non-natural amino acid 5-hydroxytryptophan, which has attracted recent interest as a novel intrinsic probe for protein
Ribosomal synthesis of cyclic peptides with a fluorogenic oxidative coupling reaction.
Yusuke Yamagishi et al.
Chembiochem : a European journal of chemical biology, 10(9), 1469-1472 (2009-05-28)
Rui Shen Ong et al.
Drug testing and analysis, 12(2), 195-214 (2019-10-09)
We describe the validation of a method for the simultaneous analysis of 29 synthetic cannabinoids (SCs) and metabolites, 4 amphetamines, and 2 cannabinoids in human whole blood. This method enables one analysis to cover what previously required multiple analyses for
Yu-Bo Wu et al.
Journal of chromatography. B, Analytical technologies in the biomedical and life sciences, 877(20-21), 1847-1855 (2009-06-02)
To make analytes amenable for fluorescence (FL) detection, polymer monolith microextraction (PMME) coupled to high-performance liquid chromatography with FL detection was developed for the simultaneous determination of catechols and 5-hydroxyindoleamines (5-HIAs) from urine samples. In this method, a two-step pre-column
Masafumi Komiya et al.
Bioorganic & medicinal chemistry, 20(23), 6840-6847 (2012-10-24)
Based on 2-(4-phenoxybenzoyl)-5-hydroxyindole (2), a novel structural class of CaMKII inhibitors were synthesized and further optimized. The strong acidity of the hydroxyl group and the lipophilic group at the 4 and 6-positions were found to be necessary for strong CaMKII
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service