134228
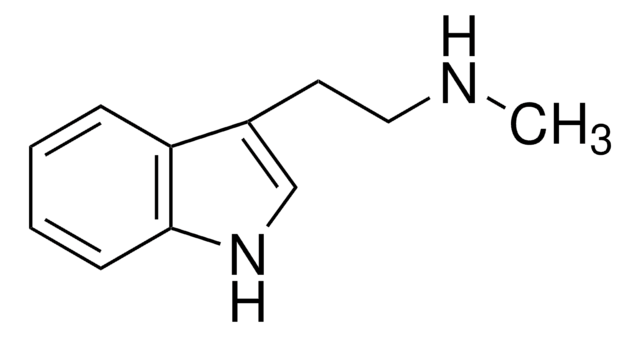
5-Methyltryptamine hydrochloride
98%
Synonym(s):
3-(2-Aminoethyl)-5-methylindole hydrochloride
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C11H14N2 · HCl
CAS Number:
Molecular Weight:
210.70
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
98%
form
powder
mp
289-292 °C (dec.) (lit.)
solubility
H2O: soluble 50 mg/mL, clear, yellow
functional group
amine
SMILES string
Cl.Cc1ccc2[nH]cc(CCN)c2c1
InChI
1S/C11H14N2.ClH/c1-8-2-3-11-10(6-8)9(4-5-12)7-13-11;/h2-3,6-7,13H,4-5,12H2,1H3;1H
InChI key
RBHDFGBPJGEYCK-UHFFFAOYSA-N
Application
5-Methyltryptamine hydrochloride was used to study the mechanism of metabolism of 9-methyl 1,2,3,4,6,7,12,12b-octahydroindolo[2,3-a]quinolizine in rats. It was used as internal standard in the determination of urinary metabolites of 5-methoxy-N,N-diisopropyltryptamine in humans. It was used as reactant in:
- synthesis of kinesin spindle protein (KSP) inhibitors
- intramolecular furan Diels-Alder reactions
- Pictet-Spengler-like reactions
- reactant in synthesis of kinesin spindle protein (KSP) inhibitors
- reactant in intramolecular furan Diels-Alder reactions
- reactant in Pictet-Spengler-like reactions
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Ruan, X., et al.
Huaxue Xuebao, 66, 1731-1731 (2008)
"Pictet-Spengler-like" Synthesis of Tetrahydro-beta-carbolines under Hydrolytic Conditions. Direct Use of Azalactones as Phenylacetaldehyde Equivalents.
James E. Audia et al.
The Journal of organic chemistry, 61(22), 7937-7939 (1996-11-01)
A Sparatore et al.
European journal of drug metabolism and pharmacokinetics, 20(2), 135-144 (1995-04-01)
The metabolic fate of 9-methyl 1,2,3,4,6,7,12,12b-octahydroindolo[2,3-a]quinolizine (MIQ), a compound with promising pharmacological action on the CNS system, was investigated in the rat after an oral dose of 200 mg/kg, the maximal tolerated dose. Urine and feces were collected, exhaustively extracted
Tooru Kamata et al.
Drug metabolism and disposition: the biological fate of chemicals, 34(2), 281-287 (2005-11-11)
The urinary metabolites of 5-methoxy-N,N-diisopropyltryptamine (5-MeO-DIPT) in humans have been investigated by analyzing urine specimens from its users. For the unequivocal identification and accurate quantification of its major metabolites, careful analyses were conducted by gas chromatography/mass spectrometry, liquid chromatography/mass spectrometry
J Sallés et al.
Brain research. Molecular brain research, 20(1-2), 111-117 (1993-10-01)
The activation of phospholipase C (PLC) was examined in membranes of rat cerebral cortex simultaneously exposed to monoaminergic receptor and muscarinic receptor agonists after the treatment of membranes with two alkylating agents, N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline (100 microM EEDQ) and propylbenzilylcholine (10 nM
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service







![1,2,3,4-Tetrahydro-9H-pyrido[3,4-b]indole 98%](/deepweb/assets/sigmaaldrich/product/structures/181/460/3d58bc34-1b5c-4295-bbac-3b52085670e8/640/3d58bc34-1b5c-4295-bbac-3b52085670e8.png)