All Photos(1)
About This Item
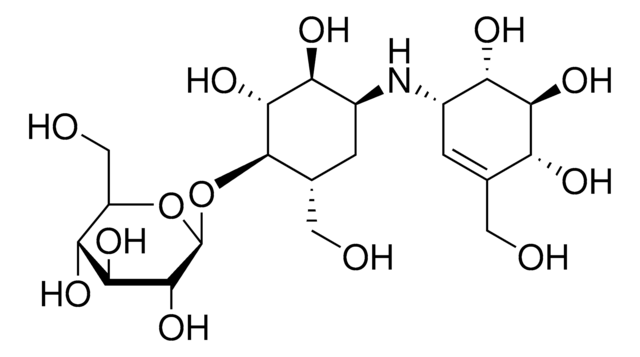
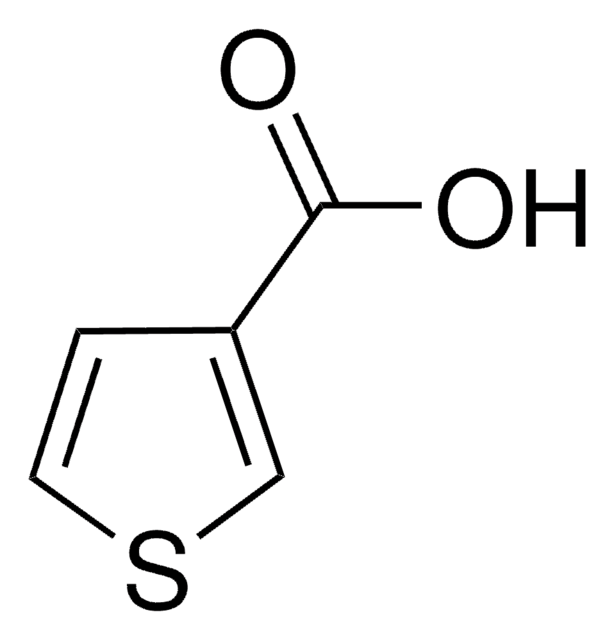
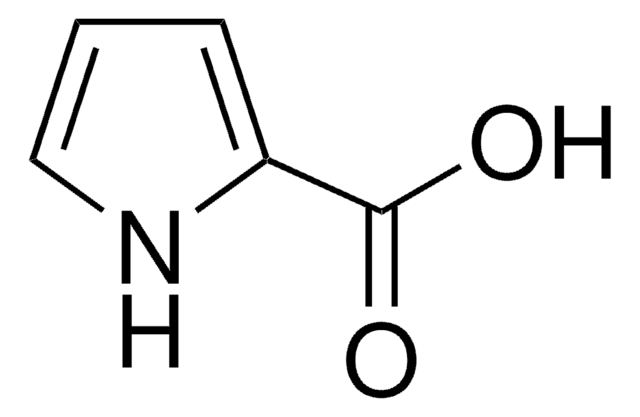
Empirical Formula (Hill Notation):
C5H4O2S
CAS Number:
Molecular Weight:
128.15
Beilstein:
110150
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
product line
ReagentPlus®
Assay
99%
bp
260 °C (lit.)
mp
125-127 °C (lit.)
SMILES string
OC(=O)c1cccs1
InChI
1S/C5H4O2S/c6-5(7)4-2-1-3-8-4/h1-3H,(H,6,7)
InChI key
QERYCTSHXKAMIS-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Legal Information
ReagentPlus is a registered trademark of Merck KGaA, Darmstadt, Germany
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Matthew F McCown et al.
Antimicrobial agents and chemotherapy, 53(5), 2129-2132 (2009-03-11)
In vitro, telaprevir selects subtype-specific resistance pathways for hepatitis C virus GT-1a and GT-1b, as described to have occurred in patients. In GT-1a, the HCV-796 resistance mutation C316Y has low replication capacity (7%) that can be compensated for by the
Hao Li et al.
Organic letters, 13(14), 3682-3685 (2011-06-17)
A stereocontrolled synthesis of α-amino-α'-alkoxy ketones is described. This pH-neutral copper(I) thiophene-2-carboxylate (CuTC)-catalyzed cross-coupling of amino acid thiol esters and chiral nonracemic α-alkoxyalkylstannanes gives α-amino-α'-alkoxy ketones in good to excellent yields with complete retention of configuration at the α-amino- and
Laval Chan et al.
Bioorganic & medicinal chemistry letters, 14(3), 797-800 (2004-01-27)
Further SAR studies on the thiophene-2-carboxylic acids are reported. These studies led to the identification of a series of tertiary amides that show inhibition of both HCV NS5B polymerase in vitro and HCV subgenomic RNA replication in Huh-7 cells. Structural
C Savarin et al.
Organic letters, 3(14), 2149-2152 (2001-07-07)
[reaction: see text] A new protocol for the palladium-catalyzed, copper-mediated coupling of aryl and alkenyl iodides with boronic acids is described. As an alternative to the well-known and widely used Suzuki cross-coupling, this reaction occurs in the absence of a
Kerim Babaoglu et al.
Nature chemical biology, 2(12), 720-723 (2006-10-31)
Fragment-based screens test multiple low-molecular weight molecules for binding to a target. Fragments often bind with low affinities but typically have better ligand efficiencies (DeltaG(bind)/heavy atom count) than traditional screening hits. This efficiency, combined with accompanying atomic-resolution structures, has made
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service


![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II)](/deepweb/assets/sigmaaldrich/product/structures/130/734/8846aa26-1858-458a-998d-8c306c13bf0f/640/8846aa26-1858-458a-998d-8c306c13bf0f.png)