All Photos(2)
About This Item
Linear Formula:
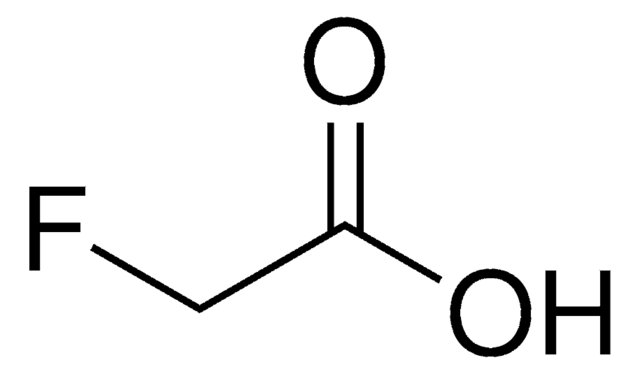
C6H5CH2CH(CH3)CO2H
CAS Number:
Molecular Weight:
164.20
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
98%
form
solid
bp
167-168 °C/23 mmHg (lit.)
mp
39-41 °C (lit.)
density
1.065 g/mL at 25 °C (lit.)
functional group
carboxylic acid
phenyl
SMILES string
CC(Cc1ccccc1)C(O)=O
InChI
1S/C10H12O2/c1-8(10(11)12)7-9-5-3-2-4-6-9/h2-6,8H,7H2,1H3,(H,11,12)
InChI key
MCIIDRLDHRQKPH-UHFFFAOYSA-N
Related Categories
General description
α-Methylhydrocinnamic acid (2-methyl-3-phenylpropionic acid, 2-benzylpropionic acid) is a cinnamic acid derivative. Its synthesis by the asymmetric hydrogenation of α-methylcinnamic acid has been reported. α-Methylhydrocinnamic acid, a short chain fatty acid derivative (SCFAD), has been reported to correct the cystic fibrosis transmembrane conductance regulator (ΔF508-CFTR) defect. Conformational studies of 2-methyl-3-phenylpropionic acid has been investigated by NMR spectroscopy. The enantiomers of 2-benzylpropionic acid has been reported to be synthesized using a lipase-catalyzed resolution. (S) (+)-2-methyl-3-phenylpropionic acid participates in the synthesis of optically active (R)-5-methyl-6-phenylhexanoyl azide. L-2-Methyl-3.phenylpropionic acid has been reported to be an inhibitor of carboxypeptidase A. Polymer-supported “Evans” oxazolidinone mediated solid phase asymmetric has been employed in the synthesis of (S)-2-methyl-3-phenylpropionic acid.
Application
α-Methylhydrocinnamic acid is suitable for use in the comparative study to investigate the γ-globin inducibility of short-chain fatty acid derivatives (SCFADs) in mice. It may be used as a histone deacetylase (HDAC) inhibitor in the comparative study to investigate the EGFP-induction potency of a number of HDAC inhibitors. It may be used in the study to investigate the selectivity of the sensor based on imprinted poly(o-phenylenediamine) (iPoPD).
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
235.4 °F - closed cup
Flash Point(C)
113 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Toan D Nguyen et al.
Biochemical and biophysical research communications, 342(1), 245-252 (2006-02-14)
Phenylalanine deletion at position 508 of the cystic fibrosis transmembrane conductance regulator (DeltaF508-CFTR), the most common mutation in cystic fibrosis (CF), causes a misfolded protein exhibiting partial chloride conductance and impaired trafficking to the plasma membrane. 4-Phenylbutyrate corrects defective DeltaF508-CFTR
M S Boosalis et al.
Blood, 97(10), 3259-3267 (2001-05-09)
Current chemotherapeutic and butyrate therapeutics that induce fetal hemoglobin expression generally also suppress erythropoiesis, limiting the production of cells containing fetal hemoglobin (F cells). Recently, selected short-chain fatty acid derivatives (SCFADs) were identified that induce endogenous gamma-globin expression in K562
Shen Li et al.
Journal of the American Chemical Society, 130(27), 8584-8585 (2008-06-13)
A highly efficient iridium-catalyzed hydrogenation of alpha,beta-unsaturated carboxylic acids has been developed by using chiral spiro-phosphino-oxazoline ligands, affording alpha-substituted chiral carboxylic acids in exceptionally high enantioselectivities and reactivities.
Stereochemical Studies. V. Intramolecular CH Bond Insertion Reaction of Acyl Nitrene generated from optically Active Acyl Azide.
Shiro T, et al.
Chemical & Pharmaceutical Bulletin, 18(6), 1124-1136 (1970)
Conformational studies of 2-methyl-3-phenylpropionic acid, 2-phenylbutyric acid and their methyl esters by NMR spectroscopy.
Spassov SL and Stefanova R.
Journal of Molecular Structure, 53, 219-224 (1979)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service