All Photos(1)
About This Item
Empirical Formula (Hill Notation):
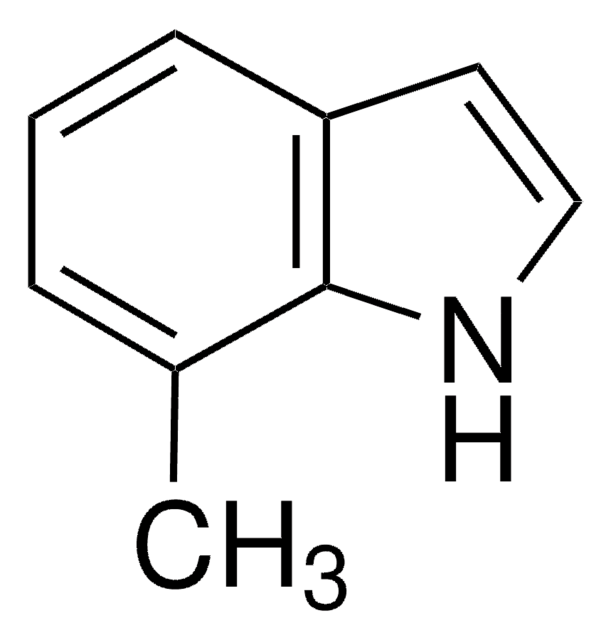
C8H6ClN
CAS Number:
Molecular Weight:
151.59
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
97%
bp
90-95 °C/0.25 mmHg (lit.)
mp
55-58 °C (lit.)
SMILES string
Clc1cccc2cc[nH]c12
InChI
1S/C8H6ClN/c9-7-3-1-2-6-4-5-10-8(6)7/h1-5,10H
InChI key
WMYQAKANKREQLM-UHFFFAOYSA-N
Related Categories
General description
7-Chloroindole is an indole derivative. It has been synthesized from 2,3-dihydroindole.
Application
7-Chloroindole may be used in the preparation of 1-methyl-7-chloroindole and glycosylated 7-chloroindole-3-acetamide.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
The chemistry of indoles. XXXIX. A facile synthetic method for 7-substituted indoles.
Somei M, et al.
Chemical & Pharmaceutical Bulletin, 35(8), 3146-3154 (1987)
Xin Teng et al.
Bioorganic & medicinal chemistry letters, 15(22), 5039-5044 (2005-09-13)
Necroptosis is a regulated caspase-independent cell death mechanism that results in morphological features resembling necrosis. It can be induced in a FADD-deficient variant of human Jurkat T cells treated with TNF-alpha. 5-(1H-Indol-3-ylmethyl)-2-thiohydantoins and 5-(1H-indol-3-ylmethyl)hydantoins were found to be potent necroptosis
Synthesis of rebeccamycin and 11-dechlororebeccamycin.
Faul MM, et al.
The Journal of Organic Chemistry, 64(7), 2465-2470 (1999)
Jeongchan Lee et al.
Nature chemical biology, 17(1), 104-112 (2020-11-04)
Tyrian purple, mainly composed of 6,6'-dibromoindigo (6BrIG), is an ancient dye extracted from sea snails and was recently demonstrated as a biocompatible semiconductor material. However, its synthesis remains limited due to uncharacterized biosynthetic pathways and the difficulty of regiospecific bromination.
Chaitany Jayprakash Raorane et al.
Biomolecules, 10(8) (2020-08-23)
Multi-drug resistant Acinetobacter baumannii is well-known for its rapid acclimatization in hospital environments. The ability of the bacterium to endure desiccation and starvation on dry surfaces for up to a month results in outbreaks of health care-associated infections. Previously, indole
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service