U211150
DMT-2′O-Methyl-rU Phosphoramidite
configured for ÄKTA® and OligoPilot®
Sinónimos:
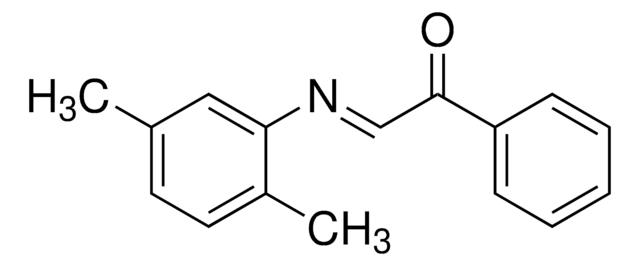
5′-O-[bis(4-methoxyphenyl)phenylmethyl]-2′-O-methyl-uridine, 3′-[2-cyanoethyl N,N-bis(1-methylethyl)phosphoramidite], DMT-2′O-Methyl-rU amidite
About This Item
Productos recomendados
biological source
non-animal source (no BSE/TSE risk)
Quality Level
product line
Proligo Reagents
assay
≥99% (31P-NMR)
≥99.0% (reversed phase HPLC)
form
powder
technique(s)
oligo synthesis: suitable
impurities
≤0.1% single unspecified Impurity (reversed phase HPLC)
≤0.3% mU2 (reversed phase HPLC, Hydrolysate)
≤0.3% mU3 (reversed phase HPLC, DMT-rme)
≤0.3% water content (Karl Fischer)
≤0.5% P(III) Impurities 100-169ppm (31P-NMR)
≤1.0% mU1 (reversed phase HPLC, DMT-rUme-DMT)
≤3% residual Solvent content
color
white to off-white
λ
conforms (UV/VIS Identity)
suitability
conforms to structure for H-NMR
conforms to structure for LC-MS
compatibility
configured for ÄKTA® and OligoPilot®
nucleoside profile
base: uridine
base protecting group: none
2' protecting group: methyl
5' protecting group: DMT
deprotection: fast/standard
storage temp.
2-8°C
SMILES string
CO[C@@H]1[C@H](OP(OCCC#N)N(C(C)C)C(C)C)[C@@H](COC(c2ccccc2)(c3ccc(OC)cc3)c4ccc(OC)cc4)O[C@H]1N5C=CC(=O)NC5=O
InChI
1S/C40H49N4O9P/c1-27(2)44(28(3)4)54(51-25-11-23-41)53-36-34(52-38(37(36)49-7)43-24-22-35(45)42-39(43)46)26-50-40(29-12-9-8-10-13-29,30-14-18-32(47-5)19-15-30)31-16-20-33(48-6)21-17-31/h8-10,12-22,24,27-28,34,36-38H,11,25-26H2,1-7H3,(H,42,45,46)/t34-,36-,37-,38-,54?/m1/s1
InChI key
UVUOJOLPNDCIHL-XKZJCBTISA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
General description
Application
Features and Benefits
- High yield of crude oligonucleotides
- Compatible with DNA synthesis
- Can be employed together with DNA or RNA phosphoramidites in the same synthesis to produce mixmer oligonucleotides
- Recommended deprotection conditions are 8 hours at 55 °C using concentrated ammonia solution, or with AMA (concentrated ammonia/ 40% aqueous methylamine I/I, v/v) for 10 minutes at 65 °C
- Purification and other downstream processing of fully modified 2′OMethyl RNA oligonucleotides are simpler than in the case of RNA, as no special precautions are required to provide protection against nucleolytic degradation
- Synthesis of 2′O-Methyl RNA oligonucleotides is similar to standard DNA synthesis but requires an elongated coupling time (recommended is 6 minutes compared to 90 seconds for DNA monomers)
- 2′O-Methyl RNA phosphoramidites are also available with fast deprotection chemistry
Other Notes
- Diagnostic probes
- Aptamer and ribozyme development
- Mixed 2′O-Methyl-RNA/DNA antisense molecules
Legal Information
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico