For information on the use of phenacyl bromide to derivatize carboxylic acids (e.g., fatty acids) for HPLC analysis, please see one or more of the following three references:1. K.J. Longmuir, et al., Determination of monoenoic fatty acid double bond position by permanganate-periodate oxidation followed by high-performance liquid chromatography of carboxylic acid phenacyl esters. Analytical Biochemistry, 167(2), 213-221 (1987).2. T. Hanis et al., Determination of fatty acids as phenacyl esters in rat adipose tissue and blood vessel walls by high-performance liquid chromatography. Journal of Chromatography, 452, 443-457 (1988). 3. A. Mehta et al., Rapid quantitation of free fatty acids in human plasma by high-performance liquid chromatography. Journal of Chromatography B Biomedical Science Applications, 719(1-2), 9-23 (1998).
77450
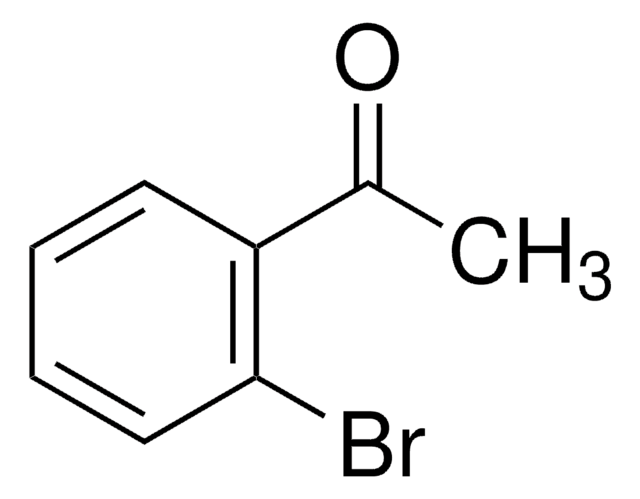
2-Bromoacetophenone
for GC derivatization, LiChropur™, ≥99.0%
Sinónimos:
ω-Bromoacetophenone, Phenacyl bromide
Seleccione un Tamaño
Seleccione un Tamaño
About This Item
Productos recomendados
grado
derivatization grade ((GC derivatization))
for GC derivatization
Nivel de calidad
Ensayo
≥99.0% (GC)
≥99.0%
Formulario
crystals
calidad
LiChropur™
idoneidad de la reacción
reagent type: derivatization reagent
reaction type: Acylations
técnicas
gas chromatography (GC): suitable
bp
135 °C/18 mmHg (lit.)
mp
48-51 °C (lit.)
49-50 °C
temp. de almacenamiento
2-8°C
cadena SMILES
BrCC(=O)c1ccccc1
InChI
1S/C8H7BrO/c9-6-8(10)7-4-2-1-3-5-7/h1-5H,6H2
Clave InChI
LIGACIXOYTUXAW-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Categorías relacionadas
Descripción general
Aplicación
Información legal
Palabra de señalización
Danger
Frases de peligro
Consejos de prudencia
Clasificaciones de peligro
Skin Corr. 1B
Código de clase de almacenamiento
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
235.4 °F - closed cup
Punto de inflamabilidad (°C)
113 °C - closed cup
Equipo de protección personal
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Elija entre una de las versiones más recientes:
Certificados de análisis (COA)
¿No ve la versión correcta?
Si necesita una versión concreta, puede buscar un certificado específico por el número de lote.
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
-
Do you have references on the use of 2-Bromoacetophenone (also known as phenacyl bromide) Products 115835 and 77450 for making phenacyl esters of carboxylic acids for HPLC analysis?
1 answer-
Helpful?
-
-
What is the Department of Transportation shipping information for this product?
1 answer-
Transportation information can be found in Section 14 of the product's (M)SDS.To access the shipping information for this material, use the link on the product detail page for the product.
Helpful?
-
Active Filters
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico