810601P
Avanti
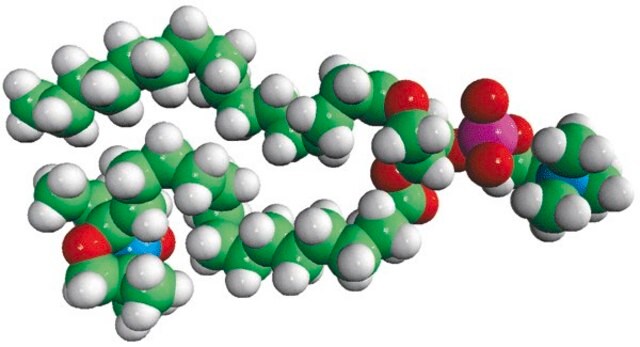
16:0-5 Doxyl PC
Avanti Research™ - A Croda Brand 810601P, powder
Synonym(s):
1-palmitoyl-2-stearoyl-(5-doxyl)-sn-glycero-3-phosphocholine
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C46H90N2O10P
CAS Number:
Molecular Weight:
862.19
UNSPSC Code:
41141825
NACRES:
NA.25
Recommended Products
Assay
>99% (TLC)
form
powder
packaging
pkg of 1 × 1 mg (810601P-1mg)
manufacturer/tradename
Avanti Research™ - A Croda Brand 810601P
lipid type
ESR probes
phospholipids
shipped in
dry ice
storage temp.
−20°C
General description
Avanti′s nitroxide spin product listing is a group of compounds designed to act as membrane probes. A variety of positions down the hydrophobic chain are labeled with the nitroxide functional groups to allow probing the membrane at various depths. These compounds have been synthesized from 1-palmitoyl-2-hydroxy-sn-glycerol-3-phosphocholine with the product being purified by column chromatography. Various n-doxyl phosphocholines have been recently used as biophysical tools to elucidate membrane trafficking with phosphatidylinositol transfer proteins and as fluorescent quenchers in lipid bilayer structural studies.
Phosphatidylcholine (PC), a strong bilayer-forming lipid is the most common phospholipid in mammalian membranes. The excretory and secretory products of helminths has a small hapten-like portion called phosphorylcholine (PC). The 5th carbon of the sn-2 stearic acid chain of 1-palmitoyl-2-stearoyl-(5-doxyl)-sn-glycero-3-phosphocholine analog has a Doxyl PC, a spin probe attached to it covalently.
Application
16:0-5 Doxyl PC may be used:
- as a component in virus-like large unilamellar vesicles (VL LUVs) to quench 4-chloro-7-nitrobenz-2-oxa-1,3-diazole (NBD) fluorescence emission
- in the preparation of multi-lamellar vesicles (MLVs) as a site-specific quencher to perform fluorescence quenching studies
- in the preparation of spin-labelled multi-lamellar vesicles (MLVs)
Biochem/physiol Actions
Phosphatidylcholine (PC) lowers the levels of cholesterol and triglycerides.
Packaging
5 mL Clear Glass Sealed Ampule (810601P-1mg)
Preparation Note
Product use: To prevent aggregation, prepare water-based solutions of 2 mM stock solutions of n-DOXYL PCs and store in plastic. Dilute stock solutions to 0.03- 0.1 mM solutions for EPR studies. For liposome preparations in fluorescent quenching measurements, dissolve the doxyl lipid in 150 μl absolute ethanol for a concentration of 40.3 mM , Additional supplemental information.
Legal Information
Avanti Research is a trademark of Avanti Polar Lipids, LLC
also commonly purchased with this product
Storage Class Code
11 - Combustible Solids
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
The Membranes of Cells (2016)
Host defenses to helminths
Clinical Immunology, 367-377 (2013)
Diego E Sastre et al.
The Journal of biological chemistry, 295(7), 2136-2147 (2019-12-05)
PlsX plays a central role in the coordination of fatty acid and phospholipid biosynthesis in Gram-positive bacteria. PlsX is a peripheral membrane acyltransferase that catalyzes the conversion of acyl-ACP to acyl-phosphate, which is in turn utilized by the polytopic membrane
Lars Duelund et al.
Biochimica et biophysica acta, 1828(8), 1909-1917 (2013-04-10)
We have in this study investigated the composition, structure and spectroscopical properties of multilamellar vesicles composed of a phospholipid, 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine (POPC), and up to 10mol% of triolein (TO), a triglyceride. We found in agreement with previous results that the mixtures
Ultra-stable temperature control in EPR experiments: thermodynamics of gel-to-liquid phase transition in spin-labeled phospholipid bilayers and bilayer perturbations by spin labels
Alaouie AM, et. al.
Journal of Magnetic Resonance, 182, 229-238 (2006)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service