All Photos(1)
About This Item
Empirical Formula (Hill Notation):
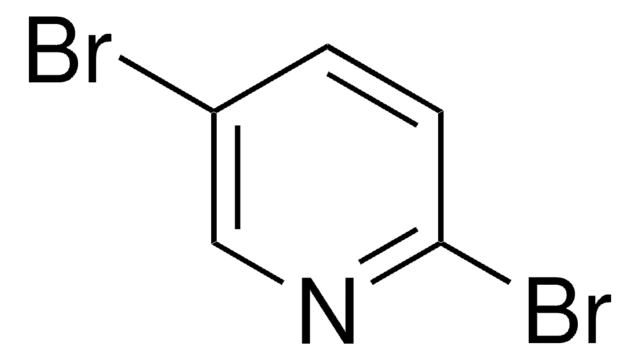
C5H3BrN2O2
CAS Number:
Molecular Weight:
202.99
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
99%
mp
148-150 °C (lit.)
functional group
bromo
nitro
SMILES string
[O-][N+](=O)c1ccc(Br)cn1
InChI
1S/C5H3BrN2O2/c6-4-1-2-5(7-3-4)8(9)10/h1-3H
InChI key
ATXXLNCPVSUCNK-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Synthesis of fluorine-18-labelled 5-and 6-fluoro-2-pyridinamine.
Abrahim A, et al.
Journal of Labelled Compounds & Radiopharmaceuticals, 49(4), 345-356 (2006)
FT-Raman and FT-IR spectra, vibrational assignments and density functional studies of 5-bromo-2-nitropyridine.
Sundaraganesan N, et al.
Spectrochimica Acta Part A: Molecular Spectroscopy, 61(13), 2995-3001 (2005)
Structure-based design and synthesis of the first weak non-phosphate inhibitors for IspF, an enzyme in the non-mevalonate pathway of isoprenoid biosynthesis.
Baumgartner C, et al.
Helvetica Chimica Acta, 90(6), 1043-1068 (2007)
Lixin Qiao et al.
Bioorganic & medicinal chemistry letters, 19(21), 6122-6126 (2009-09-29)
A structure-activity relationship study for a 2-chloroanilide derivative of pyrazolo[1,5-a]pyridine revealed that increased EphB3 kinase inhibitory activity could be accomplished by retaining the 2-chloroanilide and introducing a phenyl or small electron donating substituents to the 5-position of the pyrazolo[1,5-a]pyridine. In
Susheel J Nara et al.
The Journal of organic chemistry, 73(23), 9326-9333 (2008-11-04)
A convenient approach to 3-pyridinols and 5-pyrimidinols via a two-step Cu-catalyzed benzyloxylation/catalytic hydrogenation sequence is presented. The corresponding 3-pyridinamines and 5-pyrimidinamines can be prepared in an analogous sequence utilizing benzylamine in lieu of benzyl alcohol. The radical-scavenging ability of these
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service



![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II)](/deepweb/assets/sigmaaldrich/product/structures/130/734/8846aa26-1858-458a-998d-8c306c13bf0f/640/8846aa26-1858-458a-998d-8c306c13bf0f.png)