198005
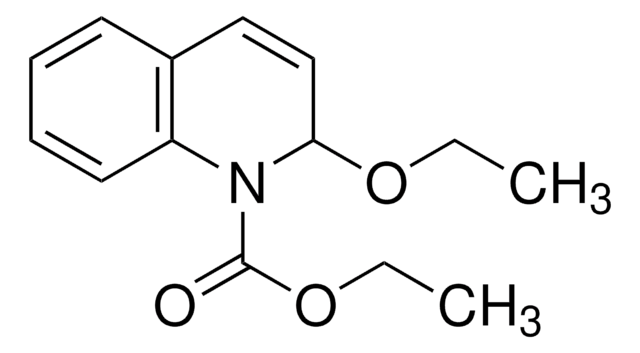
2-Chloro-1-methylpyridinium iodide
97%
Synonym(s):
Mukaiyama reagent
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Empirical Formula (Hill Notation):
C6H7ClIN
CAS Number:
Molecular Weight:
255.48
Beilstein:
3572320
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
97%
mp
200 °C (dec.) (lit.)
SMILES string
[I-].C[n+]1ccccc1Cl
InChI
1S/C6H7ClN.HI/c1-8-5-3-2-4-6(8)7;/h2-5H,1H3;1H/q+1;/p-1
InChI key
ABFPKTQEQNICFT-UHFFFAOYSA-M
Looking for similar products? Visit Product Comparison Guide
General description
2-Chloro-1-methylpyridinium iodide (Mukaiyama reagent), is a commonly used reagent in organic synthesis for the activation of the hydroxy group of alcohols and carboxylic acids. It is used to synthesize derivatives of esters, lactones, amides, lactams, and ketenes from the corresponding carboxylic acids. It is also used as an efficient coupling reagent in the synthesis of peptides because of its low toxicity, simple reaction conditions, and also less expensive than EDC.
Application
2-Chloro-1-methylpyridinium iodide (CMPI) can be used as:
- A dehydrating agent for the conversion of various aldoximes to nitriles, and alcohols into alkyl thiocyanates.
- A cross-linking agent to fabricate biodegradable cross-linked hyaluronic acid film and gelatin membranes for biomedical applications.
- A condensing agent to synthesize various β-lactams from β-amino acids.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
W Keese et al.
Biological chemistry Hoppe-Seyler, 366(12), 1093-1095 (1985-12-01)
2-Chloro-1-methylpyridinium iodide is a coupling reagent for peptide synthesis. By its use different peptides were synthesized with protected di- and trifunctional amino acids. While urethan-protected amino acids react free of racemization, the fragment condensation needs N-Hydroxysuccinimide, as shown by the
Chul-Hwan Lee et al.
Molecular cell, 70(3), 422-434 (2018-04-24)
PRC2 is a therapeutic target for several types of cancers currently undergoing clinical trials. Its activity is regulated by a positive feedback loop whereby its terminal enzymatic product, H3K27me3, is specifically recognized and bound by an aromatic cage present in
Efficient Transformation of Aldoximes to Nitriles Using 2-Chloro-1-methylpyridinium Iodide Under Mild Conditions
Lee Kieseung, et al.
Synthetic Communications, 34(10), 1775-1782 (2004)
A novel cell support membrane for skin tissue engineering: Gelatin film cross-linked with 2-chloro-1-methylpyridinium iodide
Yeh Ming-kung, et al.
Polymer, 52(4), 996-1003 (2011)
Ming-Kung Yeh et al.
Journal of biomaterials science. Polymer edition, 23(7), 973-990 (2011-05-10)
We prepared a novel porous gelatin (GEL) sponge which was cross-linked (CL) with a zero-length crosslinker of 2-chloro-1-methylpyridinium iodide (CMPI), and compared CPMI with 1-ethyl-3,3-dimethylaminoproplycarbodiimide (EDC). The ninhydrin assay indicated that the CMPI-CL-GEL sponge had a higher degree of cross-linking
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service