All Photos(1)
About This Item
Empirical Formula (Hill Notation):
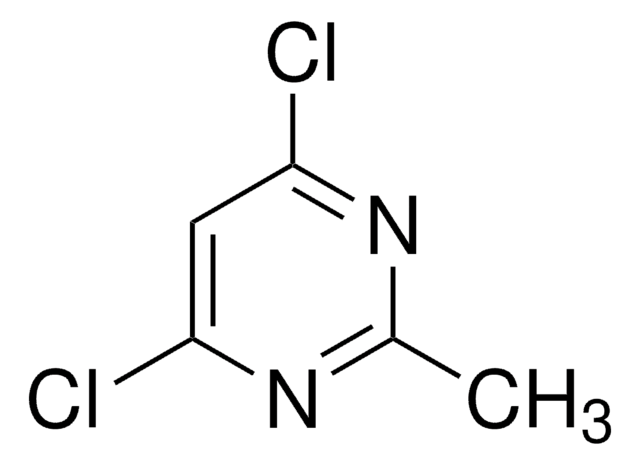
C4H2Cl2N2
CAS Number:
Molecular Weight:
148.98
Beilstein:
111195
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
97%
form
solid
bp
176 °C (lit.)
mp
65-67 °C (lit.)
solubility
95% ethanol: soluble 50 mg/mL, clear to very slightly hazy, colorless to yellow
functional group
chloro
SMILES string
Clc1cc(Cl)ncn1
InChI
1S/C4H2Cl2N2/c5-3-1-4(6)8-2-7-3/h1-2H
InChI key
XJPZKYIHCLDXST-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Cyclic voltammograms of 4,6-dichloropyrimidine shows three cathodic waves, arising from sequential cleavage of carbon-chlorine bonds as well as the reduction of pyrimidine.
Application
4,6-Dichloropyrimidine was used in the synthesis of N-substituted azacalix[4]pyrimidines. It was used as starting reagent for the synthesis of disubstituted pyrimidines by tandem amination and Suzuki-Miyaura cross-coupling. It was also used in a biarylpyrimidine synthesis involving biaryl cross-coupling.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Skin Corr. 1B
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Tetrahedron, 62, 10055-10055 (2006)
Li-Xia Wang et al.
The Journal of organic chemistry, 75(3), 741-747 (2010-01-02)
A number of N-substituted azacalix[4]pyrimidines were synthesized by two methods. While straightforward condensation reaction between 4,6-dichloropyrimidine and 4,6-bis(alkylamino)pyrimidines gave identically N-substituted azacalix[4]pyrimidines in low yields, a general and moderate-to-high yielding 1 + 3 macrocyclic fragment coupling reaction afforded azacalix[4]pyrimidines that
Richard T Wheelhouse et al.
Journal of medicinal chemistry, 49(17), 5187-5198 (2006-08-18)
Biarylpyrimidines are characterized as selective ligands for higher-order nucleic acid structures. A concise and efficient synthesis has been devised incorporating Suzuki biaryl cross-coupling of dihalopyrimidines. Two ligand series are described based on the parent thioether 4,6-bis[4-[[2-(dimethylamino)ethyl]mercapto]phenyl]pyrimidine (1a) and amide 4,6-bis(4[(2-(dimethylamino)ethyl)carboxamido]phenyl)pyrimidine
Electrochemical reduction of halogenated pyrimidines at mercury cathodes in acetonitrile.
Ji C, et al.
Journal of Electroanalytical Chemistry, 500(1), 3-11 (2001)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service



![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II)](/deepweb/assets/sigmaaldrich/product/structures/130/734/8846aa26-1858-458a-998d-8c306c13bf0f/640/8846aa26-1858-458a-998d-8c306c13bf0f.png)