852473
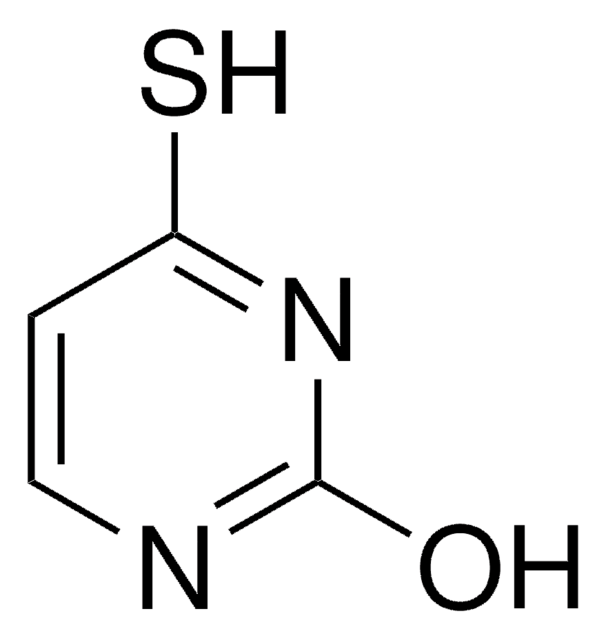
5-Bromouracil
98%
Synonym(s):
5-Bromo-2,4-dihydroxypyrimidine
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Empirical Formula (Hill Notation):
C4H3BrN2O2
CAS Number:
Molecular Weight:
190.98
Beilstein:
127176
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
98%
form
crystals
mp
>300 °C (lit.)
functional group
bromo
SMILES string
BrC1=CNC(=O)NC1=O
InChI
1S/C4H3BrN2O2/c5-2-1-6-4(9)7-3(2)8/h1H,(H2,6,7,8,9)
InChI key
LQLQRFGHAALLLE-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
The lesions produced by ultraviolet light in DNA containing 5-bromouracil.
F Hutchinson
Quarterly reviews of biophysics, 6(2), 201-246 (1973-05-01)
Victor I Danilov et al.
The journal of physical chemistry. A, 113(11), 2233-2235 (2009-02-17)
Density functional theory calculations on the canonical (keto) and rare (enol) tautomeric forms of uracil and 5-bromouracil in a cluster consisting of 50 water molecules are presented. The keto form of uracil is favored over the enol tautomer in both
Hironobu Morinaga et al.
Nucleic acids research, 41(8), 4724-4728 (2013-02-27)
Electron transfer in DNA has been intensively studied to elucidate its biological roles and for applications in bottom-up DNA nanotechnology. Recently, mechanisms of electron transfer to DNA have been investigated; however, most of the systems designed are intramolecular. Here, we
Hironobu Morinaga et al.
Bioorganic & medicinal chemistry, 21(2), 466-469 (2012-12-26)
5-Bromouracil ((Br)U) was incorporated into three types of synthetic RNA and the products of the photoirradiated (Br)U-containing RNAs were investigated using HPLC and MS analysis. The photoirradiation of r(GCA(Br)UGC)(2) and r(CGAA(Br)UUGC)/r(GCAAUUCG) in A-form RNA produced the corresponding 2'-keto adenosine ((keto)A)
Zejun Li et al.
The journal of physical chemistry. B, 115(46), 13668-13673 (2011-09-10)
The reaction of low-energy electrons (LEEs; 10 eV) with 5'-TpXpT-3' (TXT), where X is uracil (U), thymine (T), and 5-bromouracil (5BrU), was examined by HPLC-UV analysis. The presence of 5BrU increased total damage by >50%. The radiation products of T5BrUT
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service