128775
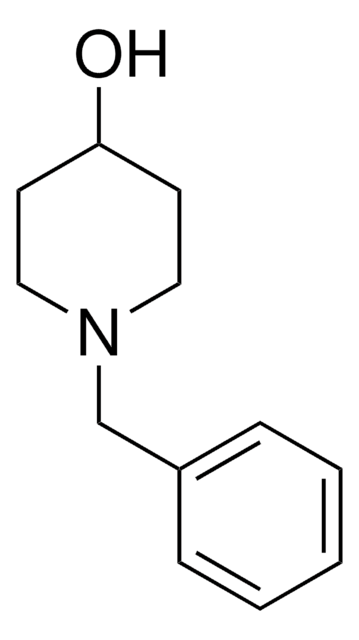
4-Hydroxypiperidine
98%
Synonym(s):
4-Piperidinol
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C5H11NO
CAS Number:
Molecular Weight:
101.15
Beilstein:
102738
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
98%
bp
108-114 °C/10 mmHg (lit.)
functional group
hydroxyl
SMILES string
OC1CCNCC1
InChI
1S/C5H11NO/c7-5-1-3-6-4-2-5/h5-7H,1-4H2
InChI key
HDOWRFHMPULYOA-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
The molecular structure, vibrational spectra, NBO and UV-spectral analysis of 4-Hydroxypiperidine have been studied. The compounds with a substituted 4-piperidinol core have been found to be potent antagonists of the human H(3) receptor.
Application
4-Hydroxypiperidine (4-Piperidinol) can be used in the synthesis of a highly potent and selective IP (PGI(2) receptor) agonist. It can also be used in the study of copper-catalyzed N- versus O-arylation.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
S Sebastian et al.
Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy, 75(3), 941-952 (2010-01-09)
In this work, we report a combined experimental and theoretical study on molecular structure, vibrational spectra, NBO and UV-spectral analysis of 4-Hydroxypiperidine (4-HP). The FT-IR solid phase (4000-400 cm(-1)), FT-IR gas phase (5000-400 cm(-1)) and FT-Raman spectra (3500-50 cm(-1)) of
N- versus O-arylation of aminoalcohols: orthogonal selectivity in copper-based catalysts.
Alexandr Shafir et al.
Journal of the American Chemical Society, 129(12), 3490-3491 (2007-03-08)
Yasuhiro Morita et al.
Molecules (Basel, Switzerland), 17(2), 1233-1246 (2012-02-02)
An efficient synthesis of a highly potent and selective IP (PGI(2) receptor) agonist that is not structurally analogous to PGI(2) is described. This synthesis is accomplished through the following key steps: Nucleophilic ring-opening of 3-(4-chlorophenyl)-oxazolidin-2-one prepared by a one-pot procedure
James T Anderson et al.
Bioorganic & medicinal chemistry letters, 20(21), 6246-6249 (2010-09-14)
Compounds containing a substituted 4-piperidinol core have been found to be potent antagonists of the human H(3) receptor. The compounds exhibited up to a 60-fold preference for inhibiting the human H(3) receptor over the mouse and showed a low binding
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service