Select a Size
| Pack Size | SKU | Availability | Price |
|---|
About This Item
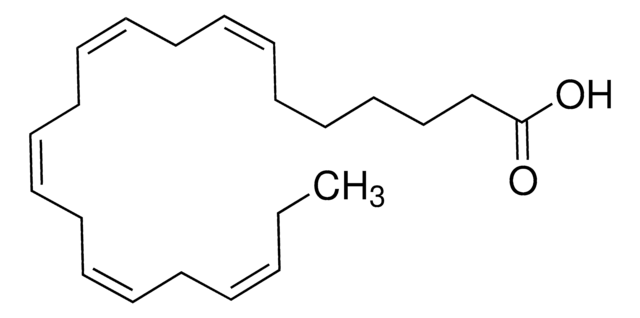
biological source
synthetic
Quality Segment
assay
≥97%
form
liquid
functional group
carboxylic acid
lipid type
unsaturated FAs
shipped in
ambient
storage temp.
−20°C
SMILES string
CC/C=C\C/C=C\C/C=C\C/C=C\C/C=C\CCCCCC(O)=O
InChI
1S/C22H34O2/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17-18-19-20-21-22(23)24/h3-4,6-7,9-10,12-13,15-16H,2,5,8,11,14,17-21H2,1H3,(H,23,24)/b4-3-,7-6-,10-9-,13-12-,16-15-
InChI key
YUFFSWGQGVEMMI-JLNKQSITSA-N
1 of 1
This Item | |||
|---|---|---|---|
| assay ≥97% | assay ≥98% (GC) | assay ≥99% | assay ≥97% |
| functional group carboxylic acid | functional group carboxylic acid | functional group carboxylic acid | functional group ester |
| biological source synthetic | biological source - | biological source synthetic (organic) | biological source - |
| form liquid | form liquid | form liquid | form liquid |
| shipped in ambient | shipped in ambient | shipped in ambient | shipped in ambient |
| storage temp. −20°C | storage temp. −20°C | storage temp. −20°C | storage temp. −20°C |
Still not finding the right product?
Explore all of our products under all-cis-7,10,13,16,19-Docosapentaenoic acid
Storage Class
10 - Combustible liquids
wgk
WGK 2
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Global Trade Item Number
| SKU | GTIN |
|---|---|
| D1797-10MG | 04061833560778 |