S2064
SC-560
≥98% (HPLC)
Synonym(s):
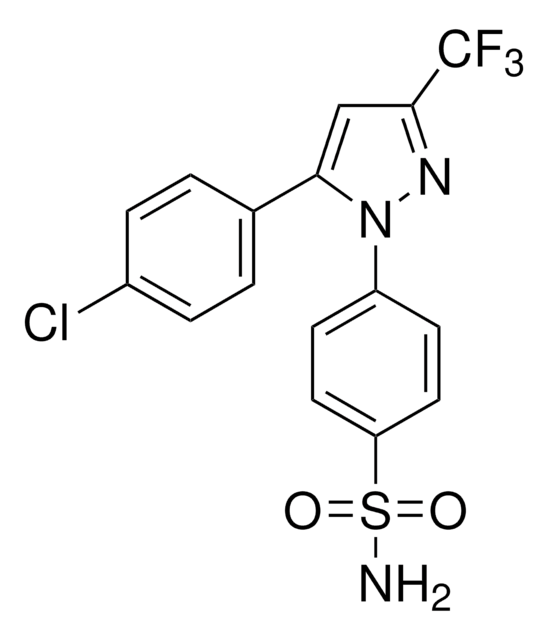
5-(4-Chlorophenyl)-1-(4-methoxyphenyl)-3-trifluoromethyl pyrazole
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C17H12ON2ClF3
CAS Number:
Molecular Weight:
352.74
MDL number:
UNSPSC Code:
51111800
PubChem Substance ID:
NACRES:
NA.77
Recommended Products
Quality Level
Assay
≥98% (HPLC)
solubility
DMSO: >20 mg/mL
storage temp.
2-8°C
SMILES string
COc1ccc(cc1)-n2nc(cc2-c3ccc(Cl)cc3)C(F)(F)F
InChI
1S/C17H12ClF3N2O/c1-24-14-8-6-13(7-9-14)23-15(10-16(22-23)17(19,20)21)11-2-4-12(18)5-3-11/h2-10H,1H3
InChI key
PQUGCKBLVKJMNT-UHFFFAOYSA-N
Gene Information
human ... PTGS1(5742) , PTGS2(5743)
Application
SC-560 has been used as a cyclooxygenase-1 (COX-1) inhibitor to study its effects on prostaglandin E-2 (PGE2) signaling in ciliogenesis in zebrafish embryos. It has also been used as a selective inhibitor of COX-1 to study its role in PM10-induced endothelial dysfunction.
Biochem/physiol Actions
SC-560 (5-(4-chlorophenyl)-1-(4-methoxyphenyl)-3-(trifluoromethyl)-1H-pyrazole) is a non-steroidal anti-inflammatory drug (NSAID). It is a lipophilic, diaryl heterocyclic compound. SC-560 acts as an effective antiviral agent against hepatitis C virus (HCV). It also has a potential to hinder prostaglandin E2 synthesis in neurons at nanomolar concentrations.
SC-560 belongs to the diaryl heterocycle class of cyclooxygenase (COX) inhibitors. It exhibits anti-tumor and anti-proliferative activities.
Selective cyclooxygenase-1 (COX-1) inhibitor, exhibiting 700-fold selectivity for COX-1 over COX-2.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
G O'Callaghan et al.
British journal of cancer, 99(3), 502-512 (2008-07-24)
Fas ligand (FasL/CD95L) is a member of the tumour necrosis factor superfamily that triggers apoptosis following crosslinking of the Fas receptor. Despite studies strongly implicating tumour-expressed FasL as a major inhibitor of the anti-tumour immune response, little is known about
Edwin S L Chan et al.
Arthritis research & therapy, 9(1), R4-R4 (2007-01-25)
Both selective cyclooxygenase (COX)-2 inhibitors and non-steroidal anti-inflammatory drugs (NSAIDs) have been beneficial pharmacological agents for many patients suffering from arthritis pain and inflammation. However, selective COX-2 inhibitors and traditional NSAIDs are both associated with heightened risk of myocardial infarction.
Dan Wang et al.
Antioxidants (Basel, Switzerland), 11(5) (2022-05-29)
Nuclear factor erythroid factor E2-related factor 2 (Nrf2) transcribes antioxidant genes that reduce the blood pressure (BP), yet its activation with tert-butylhydroquinone (tBHQ) in mice infused with angiotensin II (Ang II) increased mean arterial pressure (MAP) over the first 4
Prostaglandin signalling regulates ciliogenesis by modulating intraflagellar transport
Jin D, et al.
Nature Cell Biology, 16(9), 841-851 (2014)
Synthesis of Celecoxib, Mavacoxib, SC-560, Fluxapyroxad, and Bixafen Enabled by Continuous Flow Reaction Modules
Britton J, et al.
European Journal of Organic Chemistry, 2017(44), 6566-6574 (2017)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service