所有图片(1)
About This Item
经验公式(希尔记法):
C9H8O2
CAS号:
分子量:
148.16
MDL號碼:
分類程式碼代碼:
12352100
PubChem物質ID:
NACRES:
NA.22
推荐产品
品質等級
化驗
95%
雜質
<3% acetone
mp
175 °C (dec.) (lit.)
SMILES 字串
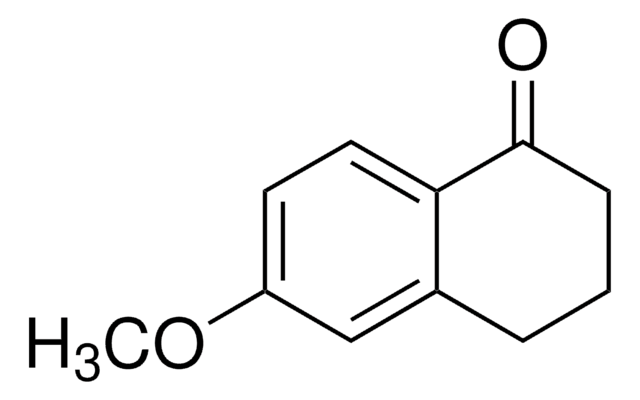
Oc1ccc2C(=O)CCc2c1
InChI
1S/C9H8O2/c10-7-2-3-8-6(5-7)1-4-9(8)11/h2-3,5,10H,1,4H2
InChI 密鑰
ZRKQOVXGDIZYDS-UHFFFAOYSA-N
一般說明
5-Hydroxy-1-indanone is a 1-indanone derivative.
應用
5-Hydroxy-1-indanone may be used in the preparation of (5-hydroxy-indan-(1E)-ylidene)-acetic acid and 5-[2-(phenyl)ethoxy]-1-indanone. It may be used as a starting material in the multi-step synthesis of 5H-indeno[1,2-c]pyridazin-5-one analogs.
訊號詞
Warning
危險聲明
危險分類
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
標靶器官
Respiratory system
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
個人防護裝備
dust mask type N95 (US), Eyeshields, Gloves
Geometric and solvent effects on intramolecular phenolic hydrogen abstraction by carbonyl n,p* and p,p* triplets.
Lathioor EC and Leigh WJ.
Canadian Journal of Chemistry, 79(12), 1851-1863 (2001)
On the involvement of single-bond rotation in the primary photochemistry of photoactive yellow protein.
Stahl AD, et al.
Biophysical Journal, 101(5), 1184-1192 (2011)
J Reniers et al.
European journal of medicinal chemistry, 46(12), 6104-6111 (2011-10-25)
Previous studies on 5H-indeno[1,2-c]pyridazin-5-one derivatives as inhibitors of MAO-B revealed that it was possible to increase the MAO-B inhibitory potency of 5H-indeno[1,2-c]pyridazin-5-ones by substituting the central heterocycle in the 3-position or C-8 with lipophilic groups which occupy the substrate cavity
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门