推荐产品
方案
≥97.0%
表单
liquid
折射率
n20/D 1.434 (lit.)
沸点
188-189 °C (lit.)
溶解性
H2O: miscible
alcohol: miscible
diethyl ether: miscible
oil: miscible
petroleum ether: miscible
密度
1.063 g/mL at 25 °C (lit.)
官能团
ether
hydroxyl
ketal
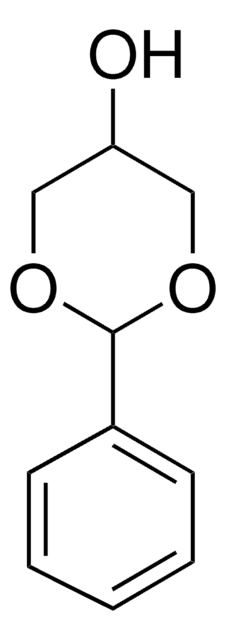
SMILES字符串
CC1(C)OCC(CO)O1
InChI
1S/C6H12O3/c1-6(2)8-4-5(3-7)9-6/h5,7H,3-4H2,1-2H3
InChI key
RNVYQYLELCKWAN-UHFFFAOYSA-N
一般描述
DL-1,2-异亚丙基甘油在磷钨酸催化下发生有效的异亚丙基化脱保护反应。
应用
以 DL-1,2-异亚丙基甘油为起始原料合成了郁金香酯衍生物。
法律信息
警示用语:
Warning
危险声明
危险分类
Eye Irrit. 2 - Repr. 2
储存分类代码
10 - Combustible liquids
WGK
WGK 1
闪点(°F)
194.0 °F - closed cup
闪点(°C)
90 °C - closed cup
个人防护装备
Eyeshields, Gloves, multi-purpose combination respirator cartridge (US)
其他客户在看
Useful methods for the synthesis of isopropylidenes and their chemoselective cleavage.
Vanlaldinpuia K and Bez G.
Tetrahedron Letters, 52(29), 3759-3764 (2011)
Alvise Perosa et al.
Molecules (Basel, Switzerland), 21(2), 170-170 (2016-02-04)
The caprylic, lauric, palmitic and stearic esters of solketal and glycerol formal were synthesized with high selectivity and in good yields by a solvent-free acid catalyzed procedure. No acetal hydrolysis was observed, notwithstanding the acidic reaction conditions.
Thomas Mendgen et al.
Bioorganic & medicinal chemistry letters, 20(19), 5757-5762 (2010-08-24)
The enzyme MurA performs an essential step in peptidoglycan biosynthesis and is therefore a target for the discovery of novel antibacterial compounds. We report here the inhibition of MurA by natural products from tulips (tulipalines and tuliposides), and the structure-activity
Enzymatic synthesis of 2,3-O-isopropylidene-sn-glycerol, a chiral building block for platelet-activating factor.
H Suemune et al.
Chemical & pharmaceutical bulletin, 34(8), 3440-3444 (1986-08-01)
P Kanda et al.
Journal of lipid research, 21(2), 257-258 (1980-02-01)
A new procedure for the preparation of 2,3-O-isopropylidene-sn-glycerol is described. L-arabinose is converted to its 4,5-monoisopropylidene diethyl mercaptal derivative. This compound is then subjected to periodate oxidation and borohydride reduction. Following neutralization, the aceton-glycerol is extracted from the aqueous solution
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门