SAE0174
MMP-2 human
recombinant, ≥1,000 pmol/min/μg, expressed in HEK 293 cells
Synonym(s):
72 kDa gelatinase, Gelatinase A, MMP-2, Matrix metalloproteinase-2, TBE-1
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
UNSPSC Code:
12352202
NACRES:
NA.32
Recommended Products
recombinant
expressed in HEK 293 cells
Quality Level
specific activity
≥1000 pmol/min-μg
shipped in
dry ice
storage temp.
−70°C
General description
Matrix Metalloproteinase-2 (MMP-2) is a member of the matrix metalloproteinase (MMP) family of proteins. MMPs participate in the breakdown of extracellular matrix in normal physiological processes like embryonic development, reproduction, and tissue remodeling, as well as in disease processes such as arthritis and metastasis. MMP-2 cleaves many substrates, including extracellular matrix components (collagens, fibronectin, and elastin), soluble metabolic mediators (e.g., apolipoproteins), secreted and extracellular matrix-anchored growth factors, and cytokines.
Along with MMP-9, MMP-2 is involved many pathophysiological processes, including leukocyte migration from the circulation into the tissue during inflammation, Chagas′ Cardiomyopathy, heart failure and chronic kidney disease. MMP-2 thus may be regarded as a potential therapeutic target.
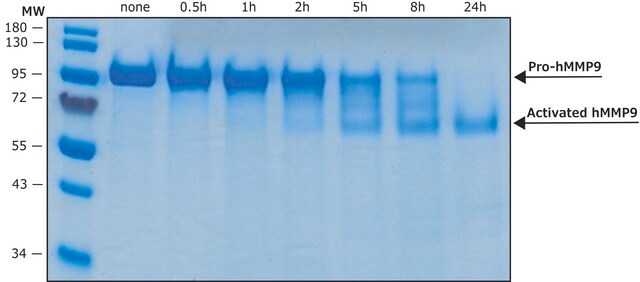
As with most MMPs, MMP-2 is secreted as an inactive pro-protein, which becomes activated when cleaved by extracellular proteinases. This product is not pre-activated, and may be activated by APMA or otherwise
This product is expressed in human HEK 293 cells as a glycoprotein with a calculated molecular mass of 72 kDa (amino acids 110-660). The DTT-reduced protein migrates as a 75-80 kDa polypeptide on SDS-PAGE because of glycosylation. This protein is produced in human cells, without the use of serum. The human cells expression system allows human like glycosylation and folding, and often supports higher specific activity of the protein. This recombinant protein is expressed without artificial tags.
Along with MMP-9, MMP-2 is involved many pathophysiological processes, including leukocyte migration from the circulation into the tissue during inflammation, Chagas′ Cardiomyopathy, heart failure and chronic kidney disease. MMP-2 thus may be regarded as a potential therapeutic target.
As with most MMPs, MMP-2 is secreted as an inactive pro-protein, which becomes activated when cleaved by extracellular proteinases. This product is not pre-activated, and may be activated by APMA or otherwise
This product is expressed in human HEK 293 cells as a glycoprotein with a calculated molecular mass of 72 kDa (amino acids 110-660). The DTT-reduced protein migrates as a 75-80 kDa polypeptide on SDS-PAGE because of glycosylation. This protein is produced in human cells, without the use of serum. The human cells expression system allows human like glycosylation and folding, and often supports higher specific activity of the protein. This recombinant protein is expressed without artificial tags.
Features and Benefits
- Highly purified protein without artificial fusion tags
- Expressed in human cells (HEK 293) for proper glycosylation
- High substrate activity after activation with APMA or other methods
Physical form
This product is supplied as a 0.22 μm-filtered solution, containing 20 mM Trizma®, pH 7.5, containing 8 mM CaCl2, 119 mM NaCl, 20% Glycerol, and 0.05% Brij® 35.
Storage and Stability
Store the product at –70 °C. The product retains its activity for at least 2 years as supplied. After initial thawing, it is recommended to store the protein in working aliquots at –70 °C.
Legal Information
Brij is a registered trademark of Croda International PLC
Trizma is a registered trademark of Merck KGaA, Darmstadt, Germany
Disclaimer
This product is for R&D use only. Not for drug, household, or other uses. Please consult the Safety Data Sheet for information regarding hazards and safe handling practices
Storage Class Code
12 - Non Combustible Liquids
WGK
WGK 1
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
M-J Hannocks et al.
Matrix biology : journal of the International Society for Matrix Biology, 75-76, 102-113 (2017-11-22)
This review focuses on the complementary roles of MMP-2 and MMP-9 in leukocyte migration into the brain in neuroinflammation, studied mainly in a murine model of experimental autoimmune encephalomyelitis (EAE) that has similarity to the human disease multiple sclerosis. We
Andrea Page-McCaw et al.
Nature reviews. Molecular cell biology, 8(3), 221-233 (2007-02-24)
Matrix metalloproteinases (MMPs) were discovered because of their role in amphibian metamorphosis, yet they have attracted more attention because of their roles in disease. Despite intensive scrutiny in vitro, in cell culture and in animal models, the normal physiological roles
Rugmani Padmanabhan Iyer et al.
American journal of physiology. Heart and circulatory physiology, 311(1), H190-H198 (2016-05-22)
Following myocardial infarction (MI), the left ventricle (LV) undergoes a series of cardiac wound healing responses that involve both the stimulation of robust inflammation to clear necrotic myocytes and tissue debris and the induction of extracellular matrix (ECM) protein synthesis
Ulrich Eckhard et al.
Matrix biology : journal of the International Society for Matrix Biology, 49, 37-60 (2015-09-27)
Secreted and membrane tethered matrix metalloproteinases (MMPs) are key homeostatic proteases regulating the extracellular signaling and structural matrix environment of cells and tissues. For drug targeting of proteases, selectivity for individual molecules is highly desired and can be met by
Nayara I Medeiros et al.
Frontiers in immunology, 10, 800-800 (2019-05-07)
Background: Chagas cardiomyopathy is the main fibrosing myocarditis among known heart diseases. Development of cardiomyopathy has been related to extracellular matrix (ECM) remodeling, which are controlled by matrix metalloproteinases (MMPs) and cytokines, especially interleukin (IL)-1β. The convertion of 31KDa inactive
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service