697400
3-Hexylthiophene-2-boronic acid pinacol ester
95%
Synonym(s):
2-(3-Hexyl-2-thienyl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane, 3-Hexyl-2-thienylboronic acid
About This Item
Recommended Products
Quality Level
Assay
95%
form
liquid
refractive index
n20/D 1.490-1.499
density
0.983 g/mL at 25 °C
SMILES string
CCCCCCc1ccsc1B2OC(C)(C)C(C)(C)O2
InChI
1S/C16H27BO2S/c1-6-7-8-9-10-13-11-12-20-14(13)17-18-15(2,3)16(4,5)19-17/h11-12H,6-10H2,1-5H3
InChI key
XCXAUPBHQCCWCI-UHFFFAOYSA-N
Application
- Suzuki-Miyaura cross-coupling reactions
- p-type/n-type switching of ambipolar bithiazole-benzothiadiazole-based polymers in solar cells
- Hierarchical self-assembly of semiconductor functionalized peptide a-helixes and optoelectronic properties
Reagent used in Preparation of
- Photovoltaic materials, polymers, and thiophene-based compounds with photophysical, electrochemical, and fluorescent properties
- Polymer solar cells for Low band gap poly(1,4-arylene-2,5-thienylene)s with benzothiadiazole units
- Dithienothiophene-based dyes for dye-sensitized solar cells
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point(F)
>230.0 °F
Flash Point(C)
> 110 °C
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
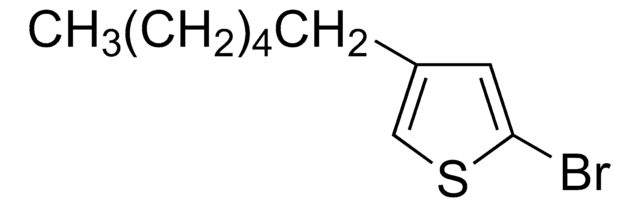
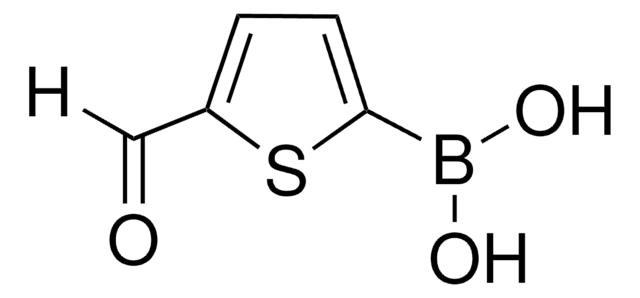
Customers Also Viewed
Articles
Oligothiophenes are important organic electronic materials which can be produced using synthetic intermediates and Suzuki coupling.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service


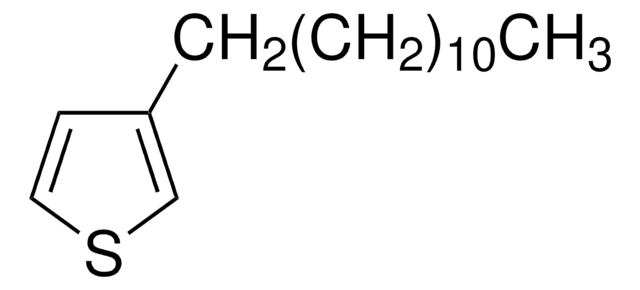








![4,7-Dibromobenzo[c]-1,2,5-thiadiazole 95%](/deepweb/assets/sigmaaldrich/product/structures/711/964/3fd3ffd1-5916-468e-a743-22f1611b5a33/640/3fd3ffd1-5916-468e-a743-22f1611b5a33.png)